How many \[\sigma \] and \[\pi \] bonds are there in the compound:
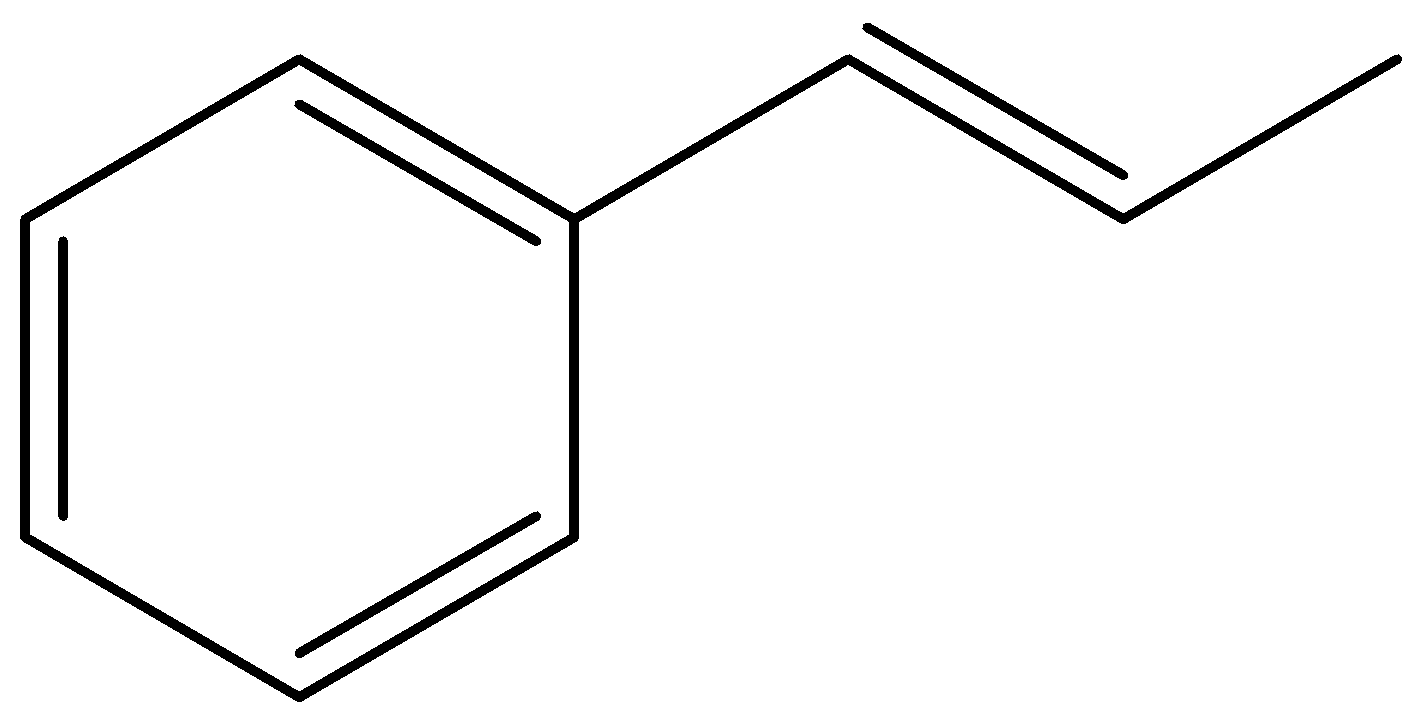
(A) 14\[\sigma \], 8\[\pi \]
(B) 18\[\sigma \],8\[\pi \]
(C) 19\[\sigma \],4\[\pi \]
(D) 14\[\sigma \],2\[\pi \]
Answer
255.6k+ views
Hint: If there is a single bond between any atom, then that bond can be considered as a \[\sigma \] bond. The multiplicity of a bond shows presence of a \[\pi \]-bond in addition to a \[\sigma \] bond.
Complete step by step solution:
A \[\pi \] bond is formed by overlap of orbitals in a side-by-side fashion and has zero electron density at its nodal planes. In other ways we can say that in a double bond, there is one \[\pi \]-bond and one \[\sigma \]-bond is present.
Let’s calculate the number of \[\pi \]-bonds present in the given compound.
You can see that there are 4 double bonds present in the compound and hence the number of \[\pi \]-bonds present in the given compound will be equal to 4.
Let’s find the number of \[\sigma \]-bonds present in the compound.
We can see that there are a total 19 \[\sigma \]-bonds present in the compound. Also draw C-H bonds and consider them in the counting of bonds as well.
So, correct answer is (C) 19\[\sigma \],4\[\pi \]
Additional Information:
A double bond is made up of one \[\sigma \]-bond and one \[\pi \]-bond. While a triple bond can be said to be made up of one \[\sigma \]-bond and two \[\pi \]-bonds.
\[\sigma \]-bond is more stronger than a \[\pi \]-bond.
Note:
Do not forget to include C-H \[\sigma \]-bonds in the counting process of total \[\sigma \]-bonds as they are often not shown in the structure and may lead to mistakes. Do not forget to include a \[\sigma \]-bond from a C-C double bond.
Complete step by step solution:
A \[\pi \] bond is formed by overlap of orbitals in a side-by-side fashion and has zero electron density at its nodal planes. In other ways we can say that in a double bond, there is one \[\pi \]-bond and one \[\sigma \]-bond is present.
Let’s calculate the number of \[\pi \]-bonds present in the given compound.
You can see that there are 4 double bonds present in the compound and hence the number of \[\pi \]-bonds present in the given compound will be equal to 4.
Let’s find the number of \[\sigma \]-bonds present in the compound.
We can see that there are a total 19 \[\sigma \]-bonds present in the compound. Also draw C-H bonds and consider them in the counting of bonds as well.
So, correct answer is (C) 19\[\sigma \],4\[\pi \]
Additional Information:
A double bond is made up of one \[\sigma \]-bond and one \[\pi \]-bond. While a triple bond can be said to be made up of one \[\sigma \]-bond and two \[\pi \]-bonds.
\[\sigma \]-bond is more stronger than a \[\pi \]-bond.
Note:
Do not forget to include C-H \[\sigma \]-bonds in the counting process of total \[\sigma \]-bonds as they are often not shown in the structure and may lead to mistakes. Do not forget to include a \[\sigma \]-bond from a C-C double bond.
Recently Updated Pages
JEE Main 2022 (June 25th Shift 2) Chemistry Question Paper with Answer Key

Average Atomic Mass - Important Concepts and Tips for JEE

JEE Main 2023 (April 6th Shift 2) Chemistry Question Paper with Answer Key

JEE Main 2022 (June 27th Shift 2) Chemistry Question Paper with Answer Key

JEE Main 2023 (January 30th Shift 2) Maths Question Paper with Answer Key

JEE Main 2022 (July 29th Shift 1) Chemistry Question Paper with Answer Key

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Understanding the Different Types of Solutions in Chemistry

Other Pages
JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

CBSE Notes Class 11 Chemistry Chapter 9 - Hydrocarbons - 2025-26

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

JEE Advanced Weightage 2025 Chapter-Wise for Physics, Maths and Chemistry

CBSE Notes Class 11 Chemistry Chapter 8 - Organic Chemistry Some Basic Principles And Techniques - 2025-26




