What is the shape of $\text{C}{{\text{H}}_{4}}$?
(a) Pyramidal
(b) Trigonal planar
(c) Linear
(d) Tetrahedral
Answer
257.1k+ views
Hint: The methods by which one can elucidate the shape of molecules are through the use of VSEPR theory. We can calculate the total bond pair of the molecule, after which geometry and shape can be predicted.
Complete step by step solution:
VSEPR stands for Valence Shell Electron Pair Repulsion theory. This theory is used to predict the shape of the molecules in a systematic way by using the number of electron pairs to determine the shape of the molecules.
Carbon is in group 4, and so has 4 outer electrons. It is forming 4 bonds to hydrogen, adding another 4 electrons-8 altogether, in 4 pairs. Because it is forming 4 bonds, these must all be bonding pairs.
Four electron pairs arrange themselves in space in what is called tetrahedral arrangement. A tetrahedron is a regular triangularly-based pyramid. The carbon atom would be at the centre and the hydrogen at the four corners. All the bond angles are ${{109.5}^{\text{o}}}$.
Shape:
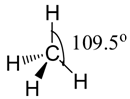
Hence, the correct option is (d) tetrahedral.
Note: It is important that you understand the use of various sorts of line to show 3-dimensional arrangement of the bonds. In diagrams of this sort, an ordinary line represents a bond in the plane of the screen or paper. A dotted line shows a bond going away from you into the screen or paper. A wedge shows a bond coming out towards you.
Complete step by step solution:
VSEPR stands for Valence Shell Electron Pair Repulsion theory. This theory is used to predict the shape of the molecules in a systematic way by using the number of electron pairs to determine the shape of the molecules.
Carbon is in group 4, and so has 4 outer electrons. It is forming 4 bonds to hydrogen, adding another 4 electrons-8 altogether, in 4 pairs. Because it is forming 4 bonds, these must all be bonding pairs.
Four electron pairs arrange themselves in space in what is called tetrahedral arrangement. A tetrahedron is a regular triangularly-based pyramid. The carbon atom would be at the centre and the hydrogen at the four corners. All the bond angles are ${{109.5}^{\text{o}}}$.
| $\text{C}{{\text{H}}_{4}}$ | |
| Central atom | Carbon |
| Valence electron on central atom | 4 |
| 4 Hydrogen atom (contributing 1 electron each) | 4 |
| Total | 8 |
| Divide by 2 | 4 |
| Shape as per VSEPR Theory | Tetrahedral |
Shape:

Hence, the correct option is (d) tetrahedral.
Note: It is important that you understand the use of various sorts of line to show 3-dimensional arrangement of the bonds. In diagrams of this sort, an ordinary line represents a bond in the plane of the screen or paper. A dotted line shows a bond going away from you into the screen or paper. A wedge shows a bond coming out towards you.
Recently Updated Pages
Electricity and Magnetism Explained: Key Concepts & Applications

JEE Energetics Important Concepts and Tips for Exam Preparation

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

JEE Main 2023 (February 1st Shift 1) Maths Question Paper with Answer Key

JEE Main 2023 (February 1st Shift 2) Maths Question Paper with Answer Key

JEE Main 2023 (February 1st Shift 2) Chemistry Question Paper with Answer Key

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

JEE Main Colleges 2026: Complete List of Participating Institutes

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Hybridisation in Chemistry – Concept, Types & Applications

Other Pages
JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

CBSE Notes Class 11 Chemistry Chapter 9 - Hydrocarbons - 2025-26

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

JEE Advanced Weightage 2025 Chapter-Wise for Physics, Maths and Chemistry

CBSE Notes Class 11 Chemistry Chapter 8 - Organic Chemistry Some Basic Principles And Techniques - 2025-26




