Pyrolysis of ethyl acetate gives:
A. \[C{H_3}COC{H_3}\]
B. \[C{H_2} = C{H_2}\]
C. \[C{H_2} = C = O\]
D. \[C{H_3} - CHO\]
Answer
266.4k+ views
Hint: Pyrolysis is the thermal decomposition of a substance at a particular temperature in an inert atmosphere, it involves a change of chemical composition. On decomposition of ester carboxylic acid and ethylene are formed.
Complete step by step answer:
Pyrolysis of ethyl acetate \[\left( {C{H_3}COOC{H_2}C{H_3}} \right)\] gives ethane and acetic acid.
\[\mathop {\left( {C{H_3}COOC{H_2}C{H_3}} \right)}\limits_{\left( {ethyl{\text{ }}acetate} \right)} \xrightarrow[{Pyrolysis}]{\Delta }\mathop {C{H_3}COOH}\limits_{\left( {acetic{\text{ }}acid} \right)} + \mathop {C{H_2} = {\text{ }}C{H_2}}\limits_{\left( {ethylene} \right)} \]
Ethyl acetate is heated in the presence of liquid nitrogen and glass wool.
Pyrolysis reaction is converting esters containing a $\beta - $hydrogen atom into the corresponding carboxylic acid and alkene. The reaction is a unimolecular elimination and operates in a syn-elimination. The mechanism involves a unimolecular six – centered transition state producing equimolar ethylene and acetic acid.
Mechanism of pyrolysis of ethyl acetate
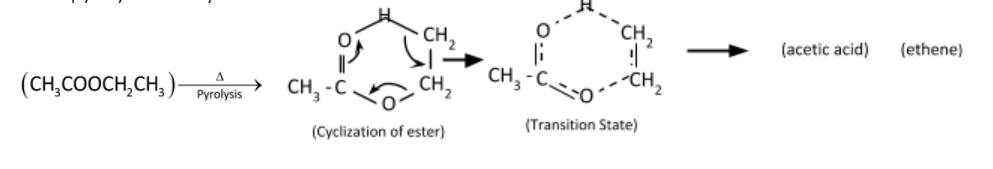
Therefore, the correct option is (D).
Note:
Ethyl acetate is a colorless liquid and is commonly known as ethyl ethanoate. Ethyl acetate is a polar aprotic solvent and it is a standard solvent. It is an important chemical compound. It is used especially for paints, varnishes, lacquers, cleaning and perfumes. The thermal decomposition of ethyl acetate follows a first order unimolecular reaction. The kinetic and thermodynamic parameters are obtained experimentally by applying the Arrhenius equation at a temperature range from $400^\circ $ to $600^\circ $C. The products from the pyrolysis of ethyl acetate were analyzed by gas chromatography.
Complete step by step answer:
Pyrolysis of ethyl acetate \[\left( {C{H_3}COOC{H_2}C{H_3}} \right)\] gives ethane and acetic acid.
\[\mathop {\left( {C{H_3}COOC{H_2}C{H_3}} \right)}\limits_{\left( {ethyl{\text{ }}acetate} \right)} \xrightarrow[{Pyrolysis}]{\Delta }\mathop {C{H_3}COOH}\limits_{\left( {acetic{\text{ }}acid} \right)} + \mathop {C{H_2} = {\text{ }}C{H_2}}\limits_{\left( {ethylene} \right)} \]
Ethyl acetate is heated in the presence of liquid nitrogen and glass wool.
Pyrolysis reaction is converting esters containing a $\beta - $hydrogen atom into the corresponding carboxylic acid and alkene. The reaction is a unimolecular elimination and operates in a syn-elimination. The mechanism involves a unimolecular six – centered transition state producing equimolar ethylene and acetic acid.
Mechanism of pyrolysis of ethyl acetate

Therefore, the correct option is (D).
Note:
Ethyl acetate is a colorless liquid and is commonly known as ethyl ethanoate. Ethyl acetate is a polar aprotic solvent and it is a standard solvent. It is an important chemical compound. It is used especially for paints, varnishes, lacquers, cleaning and perfumes. The thermal decomposition of ethyl acetate follows a first order unimolecular reaction. The kinetic and thermodynamic parameters are obtained experimentally by applying the Arrhenius equation at a temperature range from $400^\circ $ to $600^\circ $C. The products from the pyrolysis of ethyl acetate were analyzed by gas chromatography.
Recently Updated Pages
States of Matter Chapter For JEE Main Chemistry

Classification of Drugs in Chemistry: Types, Examples & Exam Guide

Types of Solutions in Chemistry: Explained Simply

Difference Between Alcohol and Phenol: Structure, Tests & Uses

[Awaiting the three content sources: Ask AI Response, Competitor 1 Content, and Competitor 2 Content. Please provide those to continue with the analysis and optimization.]

Sign up for JEE Main 2026 Live Classes - Vedantu

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




