



What Are the Key Steps of the Sun’s Fusion Process?
Nuclear fusion in the Sun is the fundamental process responsible for the production of sunlight and all forms of solar energy reaching Earth. This process involves the combination of light atomic nuclei, mainly hydrogen isotopes, under extremely high temperature and pressure conditions in the Sun’s core, leading to the release of vast amounts of energy as electromagnetic radiation.
Definition and Nature of Nuclear Fusion
Nuclear fusion refers to the reaction in which two or more light nuclei combine to form a heavier nucleus. In the context of the Sun, this process mainly involves hydrogen nuclei (protons) fusing to create helium. The mass of the resulting nucleus is slightly less than the total mass of the original nuclei, with the mass difference released as energy according to Einstein’s equation $E = \Delta m c^2$.
Fusion Reactions Occurring in the Sun
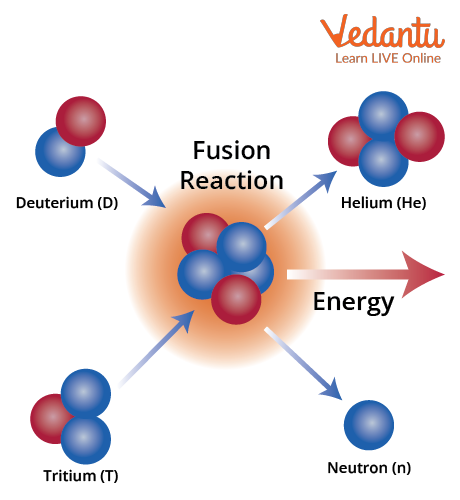
The primary nuclear fusion reaction in the Sun is known as the proton-proton chain reaction. In this sequence, four hydrogen nuclei (protons) are ultimately transformed into a single helium-4 nucleus along with the release of positrons, neutrinos, and gamma-ray photons. Each step of this chain involves overcoming the electrostatic repulsion between positively charged protons, which is achieved due to the Sun’s extreme internal temperature and pressure conditions.
The simplified overall equation for the main fusion reaction in the Sun is:
$4\, ^1\mathrm{H} \rightarrow\, ^4\mathrm{He} + 2\, \mathrm{e}^+ + 2\, \nu_e + \text{energy}$
This captures the transformation of four hydrogen nuclei into a helium nucleus, two positrons, two electron neutrinos, and a significant amount of released energy.
Proton-Proton Chain Steps
The proton-proton chain is the most prevalent fusion pathway in the Sun. The key stages of this process are as follows:
- Two protons combine to form deuterium, releasing a positron and a neutrino
- A deuterium nucleus fuses with another proton, creating helium-3 and a gamma photon
- Two helium-3 nuclei combine to form helium-4, releasing two protons
Each step requires significant energy input to overcome the Coulomb barrier, which is the repulsive force between positively charged nuclei. The energy released in these reactions sustains the Sun’s radiation and supports its equilibrium against gravitational collapse. Details about atomic structure and nucleon interactions are essential for this understanding and can be referenced in the Atomic Structure section.
Thermonuclear Conditions in the Sun’s Core
Thermonuclear fusion in the Sun requires temperatures in excess of $10^7$ Kelvin and very high pressures, achievable only in stellar cores. Under these conditions, hydrogen exists in the plasma state where electrons are dissociated from protons. This environment permits the high-energy collisions required for fusion to occur efficiently.
The Sun’s energy generation illustrates how a star maintains its structure: thermal pressure from fusion opposes gravitational collapse. These processes are addressed alongside energy principles in the Energy resource.
Energy Yield and Mass Defect
The energy produced in fusion is a direct result of the mass defect, which is the difference between the combined mass of the initial protons and the mass of the final helium nucleus. According to $E = \Delta m c^2$, this mass difference converts to energy released predominantly as photons, which gradually escape the Sun and are detected as solar radiation.
| Reaction Step | Energy Released (MeV) |
|---|---|
| $^1$H + $^1$H $\rightarrow$ $^2$H + $e^+ + \nu_e$ | 1.44 |
| $^2$H + $^1$H $\rightarrow$ $^3$He + $\gamma$ | 5.49 |
| $^3$He + $^3$He $\rightarrow$ $^4$He + 2$^1$H | 12.86 |
Solar Neutrinos and Photons
Nuclear fusion in the Sun’s core emits neutrinos and high-energy photons. Neutrinos are nearly massless and interact weakly with matter, escaping the Sun almost immediately. Photons, however, undergo numerous scatterings and can take thousands of years to reach the solar surface, eventually being emitted as sunlight.
Studies of these emitted neutrinos help confirm theoretical models of nuclear fusion in stars. Observations on this phenomenon are included in topics such as Atom And Nuclei and related nuclear interactions.
Comparison: Fusion and Fission
Nuclear fusion, such as that in the Sun, should be distinguished from nuclear fission, which is the splitting of heavy nuclei and is realized in nuclear reactors on Earth. Fusion releases significantly more energy per nucleon compared to fission. Details and comparative studies are provided in the Nuclear Fission And Fusion section.
Salient Features of Fusion Reaction in the Sun
- Operates at extremely high temperatures and pressures
- Converts hydrogen into helium via multi-step reactions
- Releases energy primarily as electromagnetic radiation
- Produces neutrinos that serve as evidence for solar fusion
- Maintains stellar equilibrium against gravitational collapse
Plasma State in Fusion Reactions
Plasma, a state of matter in which atoms are ionized, is the medium in which fusion reactions take place. The high energy environment in the Sun’s core ensures that matter is fully ionized, allowing nuclei to collide at sufficiently high rates for fusion to proceed. More about states of matter is discussed in the States Of Matter section.
Application: Solar Radiation and Earth
The energy produced by fusion in the Sun propagates outward, ultimately reaching Earth as various forms of solar radiation. This energy sustains all known life and drives atmospheric and climatic processes, as discussed in broader topics such as Gravitation and energy transfers.
FAQs on How Does Fusion Reaction Happen Inside the Sun?
1. What is the fusion reaction in the Sun?
The fusion reaction in the Sun is a nuclear process where hydrogen atoms combine to form helium, releasing immense energy. This process, known as nuclear fusion, powers the Sun and produces light and heat for the Solar System.
Key points:
- Proton-Proton Chain is the main pathway.
- Hydrogen nuclei (protons) fuse to form helium.
- Energy is released as light and heat due to mass-to-energy conversion (E = mc2).
- This energy supports life on Earth and drives solar phenomena.
2. How does nuclear fusion occur in the Sun?
Nuclear fusion occurs in the Sun when hydrogen nuclei combine at extreme temperatures and pressures in the Sun’s core.
Steps involved:
- The core’s temperature is about 15 million °C.
- High pressure and temperature allow protons to overcome repulsive forces.
- Two protons fuse to form deuterium (a hydrogen isotope).
- Further reactions form helium-4 and release energy, positrons, and neutrinos.
- This energy radiates outward as sunlight.
3. What is the proton-proton chain reaction in the Sun?
The proton-proton chain reaction is the main fusion process in the Sun converting hydrogen into helium.
Main steps:
- Two protons fuse to form deuterium.
- Deuterium fuses with another proton to produce helium-3.
- Two helium-3 nuclei combine to form helium-4 and release protons.
- The process releases energy in the form of gamma rays, positrons, and neutrinos.
4. Why is fusion important for the Sun?
Fusion is crucial for the Sun because it is the source of the Sun’s energy and stability.
Importance of fusion:
- Produces the light and heat needed for life on Earth.
- Generates solar radiation that drives weather and climate.
- Keeps the Sun stable by balancing gravitational collapse with energy output.
- Ensures the Sun’s long lifespan by steadily converting hydrogen to helium.
5. What conditions are necessary for fusion to occur in the Sun?
Fusion requires extremely high temperature and pressure to occur in the Sun’s core.
Key conditions include:
- Core temperature of about 15 million °C (27 million °F).
- Immense pressure from the Sun’s gravity.
- High density of hydrogen nuclei for frequent collisions.
These extreme conditions make nuclear fusion possible in the Sun but difficult to achieve on Earth.
6. How does fusion in the Sun differ from fission in nuclear reactors?
Fusion and fission are opposite nuclear processes but both release large amounts of energy.
Differences:
- Fusion: Light nuclei (hydrogen) combine to form heavier nuclei (helium).
- Fission: Heavy nuclei (uranium, plutonium) split into lighter nuclei.
- Fusion occurs in the Sun’s core under high temperature and pressure, while fission occurs in nuclear reactors on Earth at lower temperatures.
- Fusion produces less radioactive waste compared to fission.
7. What is the balanced nuclear equation for the fusion reaction in the Sun?
The balanced equation for hydrogen fusion in the Sun is:
41H → 4He + 2e+ + 2ν + energy
Where:
- Four hydrogen nuclei fuse to form one helium nucleus.
- Two positrons (e+) and two neutrinos (ν) are released.
- Energy is released according to Einstein’s equation (E = mc2).
8. Why does mass decrease during nuclear fusion in the Sun?
Mass decreases during fusion because some mass of the reactants is converted to energy.
Main points:
- Initial mass of hydrogen nuclei is slightly more than the final helium nucleus.
- The difference in mass is called the mass defect.
- This mass defect is converted into energy as per E = mc2.
This released energy powers the Sun and reaches us as solar radiation.
9. How does energy from fusion reach the Earth?
Energy from fusion in the Sun travels to Earth mainly as solar radiation.
Energy transfer steps:
- Energy produced in the core moves outward by radiation and convection within the Sun.
- Reaches the Sun’s surface and is emitted as light (visible rays) and heat (infrared rays).
- Travels through space and reaches Earth in about 8 minutes.
10. What are the by-products of fusion in the Sun?
The by-products of fusion in the Sun are energy, helium nuclei, positrons, and neutrinos.
Details:
- Helium-4 nuclei (main product)
- Energy (in the form of photons)
- Positrons (e+), which annihilate with electrons
- Neutrinos (ν), which escape the Sun almost undisturbed
11. What is the role of temperature and pressure in fusion reactions inside the Sun?
High temperature and pressure in the Sun’s core are vital for fusion to occur.
Key points:
- High temperature gives protons enough energy to overcome repulsive forces.
- Pressure squeezes nuclei closer together for collisions.
- These extreme conditions are sustained by the Sun’s massive gravitational force.
12. How long will the Sun continue to produce energy through fusion?
The Sun will produce energy through fusion for an estimated total of about 10 billion years.
- The Sun is currently around 4.6 billion years old.
- It has enough hydrogen fuel to sustain fusion for about another 5 billion years.
- After that, it will evolve into a red giant and eventually end its life cycle.































