Ortho-hydrogen and para-hydrogen resembles in which of the following property
A. Thermal conductivity
B. Magnetic properties
C. Chemical properties
D. Heat capacity
Answer
259.5k+ views
Hint: Ortho- hydrogen has parallel nuclear spins whereas para- hydrogen has Anti parallel spins. Due to having two parallel spins, ortho- hydrogen has an overall nuclear spin of $1$ and para-hydrogen has an overall nuclear spin-zero. Ortho-hydrogen and parahydrogen both have different physical properties.
Complete Step by Step Answer:
The hydrogen (H) atoms have one proton inside the nucleus and one electron rotates around it. Proton also spins around its axis like an electron. If the protons of two H atoms of ${{H}_{2}}$ molecule rotate in the same direction then they are called ortho-hydrogen. When the protons of two hydrogens rotate in opposite directions then they are called para-hydrogen. Ortho and para-hydrogen are spin isomers of the molecular dihydrogen. At ${{0}^{{\mathrm O}}}K$hydrogen consists mainly of para-hydrogen and para-hydrogen is more stable than ortho-hydrogen. Ordinary hydrogen is an equilibrium mixture of both ortho and para hydrogen.
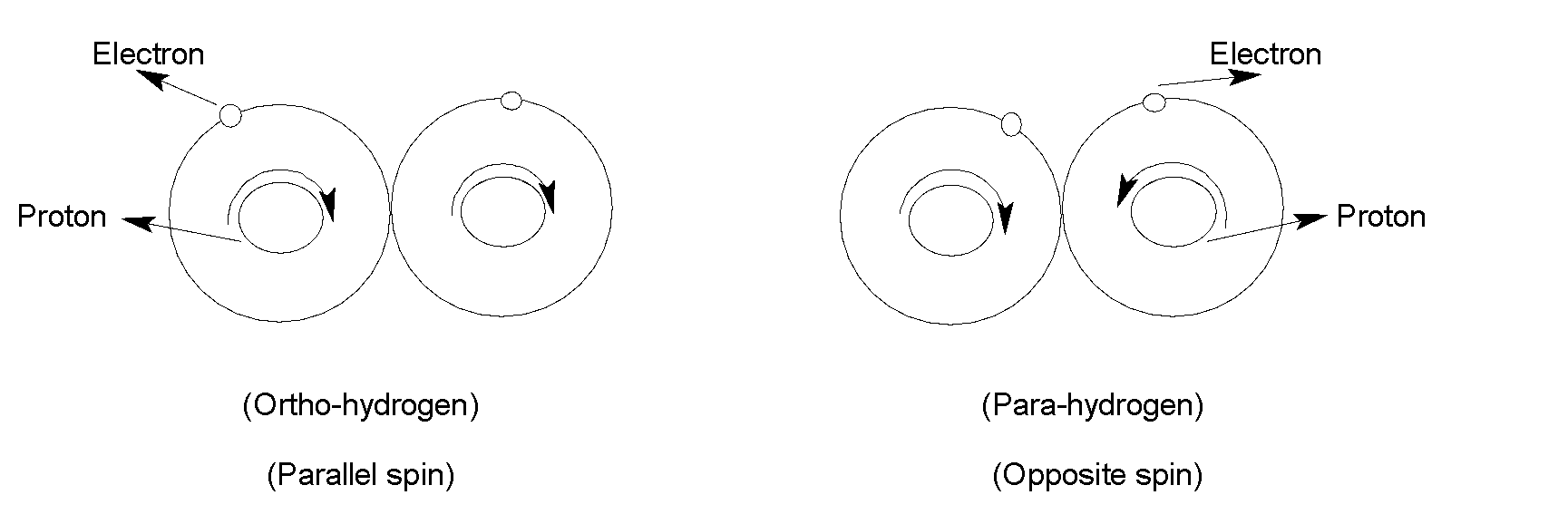
Ortho-hydrogen has an overall spin$=\left( +\frac{1}{2}+\frac{1}{2} \right)=1$
And para-hydrogen has an overall spin $=\left( +\frac{1}{2}-\frac{1}{2} \right)=0$
That is ortho hydrogen is in a triplet state as its overall nuclear spin is $1$ and para-hydrogen is in a singlet state as it has zero nuclear spins.
They have different nuclear energies due to differences in an overall nuclear spin. For this property, ortho-H and para-H have different physical properties such as thermal conductivity, heat capacity, boiling point, etc. But they have similar chemical properties because their electronic configuration is similar.
Thus, option (C) is correct.
Note: Para hydrogen can be converted into ortho-hydrogen in many different ways such as by passing an electric charge, by heating at a temperature greater than ${{800}^{{\mathrm O}}}C$, by treating with catalysis iron or platinum metal, by mixing with paramagnetic molecules such as NO, ${{O}_{2}}$ etc.
Complete Step by Step Answer:
The hydrogen (H) atoms have one proton inside the nucleus and one electron rotates around it. Proton also spins around its axis like an electron. If the protons of two H atoms of ${{H}_{2}}$ molecule rotate in the same direction then they are called ortho-hydrogen. When the protons of two hydrogens rotate in opposite directions then they are called para-hydrogen. Ortho and para-hydrogen are spin isomers of the molecular dihydrogen. At ${{0}^{{\mathrm O}}}K$hydrogen consists mainly of para-hydrogen and para-hydrogen is more stable than ortho-hydrogen. Ordinary hydrogen is an equilibrium mixture of both ortho and para hydrogen.

Ortho-hydrogen has an overall spin$=\left( +\frac{1}{2}+\frac{1}{2} \right)=1$
And para-hydrogen has an overall spin $=\left( +\frac{1}{2}-\frac{1}{2} \right)=0$
That is ortho hydrogen is in a triplet state as its overall nuclear spin is $1$ and para-hydrogen is in a singlet state as it has zero nuclear spins.
They have different nuclear energies due to differences in an overall nuclear spin. For this property, ortho-H and para-H have different physical properties such as thermal conductivity, heat capacity, boiling point, etc. But they have similar chemical properties because their electronic configuration is similar.
Thus, option (C) is correct.
Note: Para hydrogen can be converted into ortho-hydrogen in many different ways such as by passing an electric charge, by heating at a temperature greater than ${{800}^{{\mathrm O}}}C$, by treating with catalysis iron or platinum metal, by mixing with paramagnetic molecules such as NO, ${{O}_{2}}$ etc.
Recently Updated Pages
Disproportionation Reaction: Definition, Example & JEE Guide

Hess Law of Constant Heat Summation: Definition, Formula & Applications

JEE General Topics in Chemistry Important Concepts and Tips

JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Understanding the Different Types of Solutions in Chemistry

Other Pages
JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

JEE Advanced Weightage 2025 Chapter-Wise for Physics, Maths and Chemistry

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners




