How is nitromethane prepared from
(i) $\alpha - $chloro sodium acetate
(ii) $\alpha - $nitro isobutylene?
Answer
255.9k+ views
Hint: Nitroalkanes can be obtained by treating alkyl halides with silver nitrate in alcoholic solution. Hydrolysis of $\alpha - $nitro alkene can produce nitromethane.
Complete step by step solution:
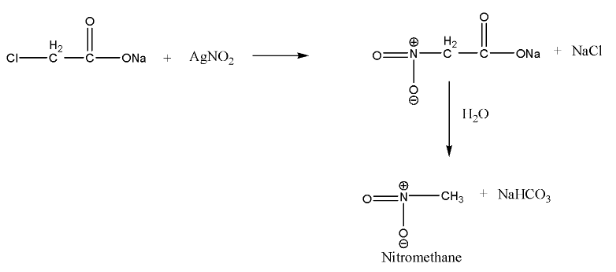
When $\alpha-$ chloro sodium acetate is treated with silver nitrate, the chlorine gets replaced by the nitro group and $\alpha - $nitro sodium acetate is formed, which undergoes hydrolysis to give nitro methane and sodium bicarbonate as a by-product.
This reaction is a nucleophilic substitution reaction. Since silver nitrite is predominantly covalent, only nitrogen pairs are available for bond formation. Hence, on adding $AgN{O_2}$, the attack of $NO_2^- $ takes place mainly through nitrogen and a nitroalkane is obtained as the major product.
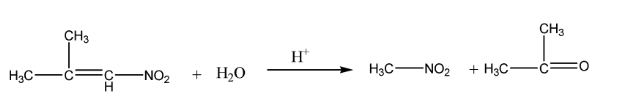
$\alpha - $nitro isobutylene undergoes hydrolysis in an acidic medium to give nitromethane and acetone.
Note: When $\alpha - $ chloro sodium acetate is treated with silver nitrate, some alkyl nitrate is also formed ($20-30\% $) in the reaction along with nitroalkane. This is because$NO_2^-$ ion is an ambident nucleophile and can attack the alkyl halide through nitrogen as well as through oxygen. If $\alpha -$ chloro sodium acetate is treated with $NaN{O_2}$ or $KN{O_2}$ in place of $AgN{O_2}$, the main product will be alkyl nitrite. Nitroalkenes do not undergo hydrolysis in the basic medium to form nitromethane.
Complete step by step solution:
When $\alpha-$ chloro sodium acetate is treated with silver nitrate, the chlorine gets replaced by the nitro group and $\alpha - $nitro sodium acetate is formed, which undergoes hydrolysis to give nitro methane and sodium bicarbonate as a by-product.
This reaction is a nucleophilic substitution reaction. Since silver nitrite is predominantly covalent, only nitrogen pairs are available for bond formation. Hence, on adding $AgN{O_2}$, the attack of $NO_2^- $ takes place mainly through nitrogen and a nitroalkane is obtained as the major product.

$\alpha - $nitro isobutylene undergoes hydrolysis in an acidic medium to give nitromethane and acetone.

Note: When $\alpha - $ chloro sodium acetate is treated with silver nitrate, some alkyl nitrate is also formed ($20-30\% $) in the reaction along with nitroalkane. This is because$NO_2^-$ ion is an ambident nucleophile and can attack the alkyl halide through nitrogen as well as through oxygen. If $\alpha -$ chloro sodium acetate is treated with $NaN{O_2}$ or $KN{O_2}$ in place of $AgN{O_2}$, the main product will be alkyl nitrite. Nitroalkenes do not undergo hydrolysis in the basic medium to form nitromethane.
Recently Updated Pages
Electricity and Magnetism Explained: Key Concepts & Applications

JEE Energetics Important Concepts and Tips for Exam Preparation

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

JEE Main 2023 (February 1st Shift 1) Maths Question Paper with Answer Key

JEE Main 2023 (February 1st Shift 2) Maths Question Paper with Answer Key

JEE Main 2023 (February 1st Shift 2) Chemistry Question Paper with Answer Key

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

JEE Main Colleges 2026: Complete List of Participating Institutes

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Hybridisation in Chemistry – Concept, Types & Applications

Other Pages
JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

CBSE Notes Class 11 Chemistry Chapter 9 - Hydrocarbons - 2025-26

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

JEE Advanced Weightage 2025 Chapter-Wise for Physics, Maths and Chemistry

CBSE Notes Class 11 Chemistry Chapter 8 - Organic Chemistry Some Basic Principles And Techniques - 2025-26




