Main Products formed during a reaction of 1-methoxy naphthalene with hydroiodic acid are:
A.
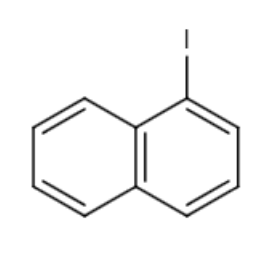 and CH3I
and CH3I
B.
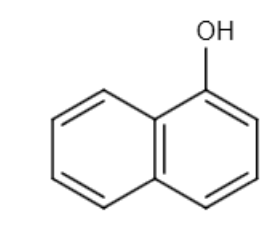 and CH3OH
and CH3OH
C.
 and CH3OH
and CH3OH
D.
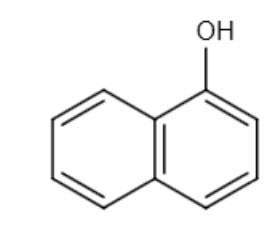 and CH3I
and CH3I
Answer
260.1k+ views
Hint: Here, in this question, 1-methoxy naphthalene is given. It reacts with hydroiodic acid. First of all, hydrogen ions react with 1-methoxy naphthalene followed by iodide ions.
Complete Step by Step Solution:
The structure of 1-methoxy naphthalene is as follows:
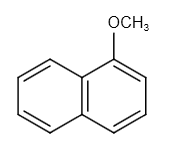
1-methoxy naphthalene reacts with hydrogen ion as follows:
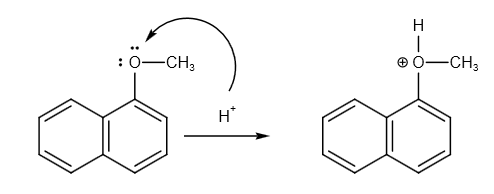
Hydrogen ions attack oxygen and bind with oxygen.
It will further react with iodine ion as follows:
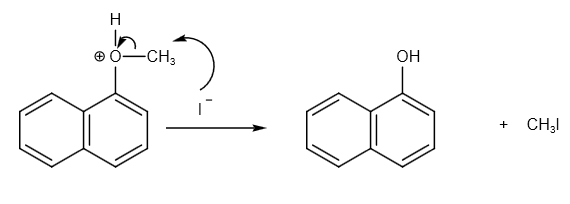
The carbon and oxygen bond breaks and iodine ions attack carbon to form methyl iodide.
Therefore, the correct answer is option D.
Additional Information:
The odour of naphthalene, a white, crystalline volatile substance, is often compared to that of mothballs. The substance slowly sublimes (changes from a solid to a gas) at room temperature, producing a vapour that is extremely combustible. The structure of naphthalene can be used to predict how soluble it is in water. A polyatomic hydrocarbon is naphthene. It is a hydrophobic molecule since it contains a lot of carbon atoms. The hydrophobic nature of this naphthalene makes it insoluble in water. The liquid nature of naphthalene is not thoroughly covered in this article. In 1-methoxy naphthalene, there is one hydrogen of naphthalene substituted by the methyl group.
Note: We must keep in mind that hydrogen iodide is a halide of hydrogen and a diatomic molecule. Aqueous solutions of HI are also referred to as hydroiodic acid, a solid acid. Hydroiodic acid and hydrogen iodide, however, are different in that the former is a gas under normal circumstances while the latter is an aqueous gas solution.
Complete Step by Step Solution:
The structure of 1-methoxy naphthalene is as follows:

1-methoxy naphthalene reacts with hydrogen ion as follows:

Hydrogen ions attack oxygen and bind with oxygen.
It will further react with iodine ion as follows:

The carbon and oxygen bond breaks and iodine ions attack carbon to form methyl iodide.
Therefore, the correct answer is option D.
Additional Information:
The odour of naphthalene, a white, crystalline volatile substance, is often compared to that of mothballs. The substance slowly sublimes (changes from a solid to a gas) at room temperature, producing a vapour that is extremely combustible. The structure of naphthalene can be used to predict how soluble it is in water. A polyatomic hydrocarbon is naphthene. It is a hydrophobic molecule since it contains a lot of carbon atoms. The hydrophobic nature of this naphthalene makes it insoluble in water. The liquid nature of naphthalene is not thoroughly covered in this article. In 1-methoxy naphthalene, there is one hydrogen of naphthalene substituted by the methyl group.
Note: We must keep in mind that hydrogen iodide is a halide of hydrogen and a diatomic molecule. Aqueous solutions of HI are also referred to as hydroiodic acid, a solid acid. Hydroiodic acid and hydrogen iodide, however, are different in that the former is a gas under normal circumstances while the latter is an aqueous gas solution.
Recently Updated Pages
Algebra Made Easy: Step-by-Step Guide for Students

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

JEE Energetics Important Concepts and Tips for Exam Preparation

Chemical Properties of Hydrogen - Important Concepts for JEE Exam Preparation

JEE General Topics in Chemistry Important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Understanding the Different Types of Solutions in Chemistry

Derivation of Equation of Trajectory Explained for Students

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

Understanding Atomic Structure for Beginners

How to Convert a Galvanometer into an Ammeter or Voltmeter




