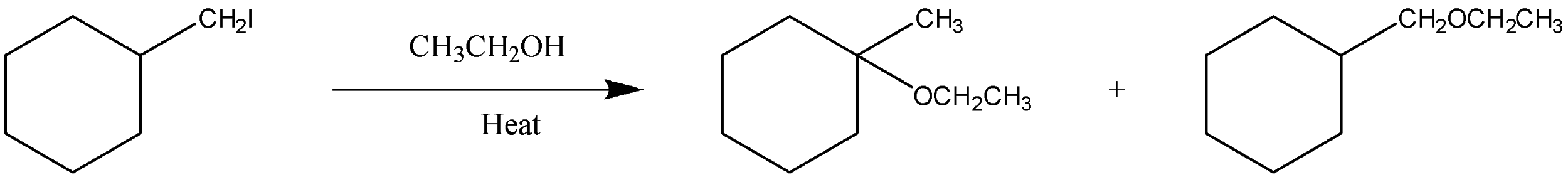
Is the above reaction an example of solvolysis reaction? Give the reason for your answer.
Answer
262.2k+ views
Hint: Solvolysis reactions are kind of substitution reactions. In these reactions solvent displaces an atom present in a molecule. The ethanol given here is a solvent molecule.
Complete step by step answer:
* First, let us know about the solvolysis, or solvolytic reactions. In this reaction an atom, or group present in a molecule is replaced by another atom, or group of atoms. The solvent present in the reaction produces electron-rich atoms that act as a nucleophile, and displace an atom.
* Now, in the given reaction ethanol present behaves as a solvent as mentioned, so it will act as a nucleophile too.
* The first step will be the formation of a carbocation by displacement of an atom I present at the ortho position, we can see it in the figure.
* In the second step, nucleophile formed will attack, and then there will be the deprotonation of molecules.
Thus, we can say that the end product given in the figure will be formed after the deprotonation of a molecule.
So, this mechanism, and the reaction given satisfies all the conditions of solvolysis reaction.
Hence, we can conclude that the given reaction is an example of solvolysis reaction.
Note: Don’t get confused while identifying the reaction, we can clearly see that there is presence of ethanol which is a solvent, and an atom is also present to displace, and form a carbocation. So, at last there is removal of a proton. Just do it step by step, and we would know the answer.
Complete step by step answer:
* First, let us know about the solvolysis, or solvolytic reactions. In this reaction an atom, or group present in a molecule is replaced by another atom, or group of atoms. The solvent present in the reaction produces electron-rich atoms that act as a nucleophile, and displace an atom.
* Now, in the given reaction ethanol present behaves as a solvent as mentioned, so it will act as a nucleophile too.
* The first step will be the formation of a carbocation by displacement of an atom I present at the ortho position, we can see it in the figure.
* In the second step, nucleophile formed will attack, and then there will be the deprotonation of molecules.
Thus, we can say that the end product given in the figure will be formed after the deprotonation of a molecule.
So, this mechanism, and the reaction given satisfies all the conditions of solvolysis reaction.
Hence, we can conclude that the given reaction is an example of solvolysis reaction.
Note: Don’t get confused while identifying the reaction, we can clearly see that there is presence of ethanol which is a solvent, and an atom is also present to displace, and form a carbocation. So, at last there is removal of a proton. Just do it step by step, and we would know the answer.
Recently Updated Pages
JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Electricity and Magnetism Explained: Key Concepts & Applications

JEE Energetics Important Concepts and Tips for Exam Preparation

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Understanding the Different Types of Solutions in Chemistry

Derivation of Equation of Trajectory Explained for Students

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




