In the reaction ${C_6}{H_6}\xrightarrow{{C{H_3}Cl/AlC{l_3}}}A\xrightarrow{{KMn{O_4}}}B$, B is :
a.) benzoic acid
b.) benzoyl chloride
c.) benzaldehyde
d.) chlorobenzene
Answer
257.4k+ views
Hint: The first reagent given is the methyl chloride in the presence of aluminium trichloride. It functions in alkylation of the compound while the second reagent functions in oxidation of the compound. The answer to this question is a carboxylic acid with molecular mass 122 g. This is because the ultimate product of oxidation of alkyl is the acid.
Complete step by step solution:
Let us move step by step in the reaction to get the correct product.
We have the initial reactant benzene. We know that benzene is electron rich in nature. It is even highly stable due to resonance and thus it does not let resonance chain break. So, it does not give addition reactions.
It can very easily give aromatic electrophilic substitution reactions.
The first reagent is the methyl chloride in the presence of aluminium trichloride. It is an alkylation reaction where benzene gets converted into toluene and HCl is released as a byproduct. The A product formed is toluene. This reaction is Friedel Crafts alkylation. The reaction can be written as -
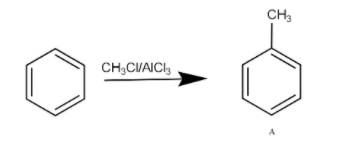
This reaction involves following mechanism-
The methyl chloride reacts with aluminium chloride to give $ \oplus C{H_3}$carbocation. This methyl carbocation is an electrophile and this gets attached to benzene.
$C{H_3}Cl + AlC{l_3} \to \oplus C{H_3}$

This carbocation is stabilised by resonance as-
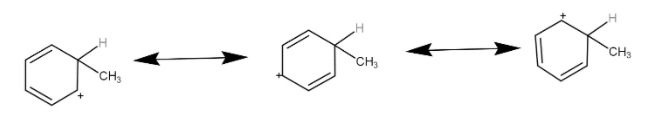
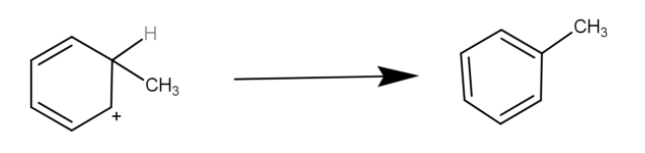
The hydrogen is eliminated restoring resonance.
The second reagent that acts on toluene is potassium permanganate. The potassium permanganate is used for oxidation and here also, it does oxidation of the methyl group of toluene.
The oxidation is the addition of oxygen or removal of hydrogen. Here, the oxygen will get bonded with carbon and result in the formation of Benzoic acid. The benzoic acid is the product B.
The answer can-not be benzaldehyde because the potassium permanganate is a very strong reagent and it converts the benzaldehyde formed into benzoic acid. Firstly, from toluene benzaldehyde is formed which later gets converted into benzoic acid.
The reaction for this is as -
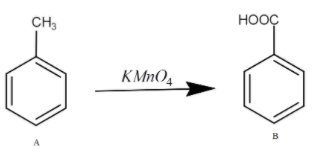
So, the answer is option a.)
Note: It must be noted that alkylation means the addition of alkyl groups. As here, the reagent was methyl chloride; this means the reagent had a methyl group. So, the methylation occurred. If it would have been ethyl chloride along with aluminium trichloride, the ethylation would have occurred.
Complete step by step solution:
Let us move step by step in the reaction to get the correct product.
We have the initial reactant benzene. We know that benzene is electron rich in nature. It is even highly stable due to resonance and thus it does not let resonance chain break. So, it does not give addition reactions.
It can very easily give aromatic electrophilic substitution reactions.
The first reagent is the methyl chloride in the presence of aluminium trichloride. It is an alkylation reaction where benzene gets converted into toluene and HCl is released as a byproduct. The A product formed is toluene. This reaction is Friedel Crafts alkylation. The reaction can be written as -

This reaction involves following mechanism-
The methyl chloride reacts with aluminium chloride to give $ \oplus C{H_3}$carbocation. This methyl carbocation is an electrophile and this gets attached to benzene.
$C{H_3}Cl + AlC{l_3} \to \oplus C{H_3}$

This carbocation is stabilised by resonance as-

The hydrogen is eliminated restoring resonance.

The second reagent that acts on toluene is potassium permanganate. The potassium permanganate is used for oxidation and here also, it does oxidation of the methyl group of toluene.
The oxidation is the addition of oxygen or removal of hydrogen. Here, the oxygen will get bonded with carbon and result in the formation of Benzoic acid. The benzoic acid is the product B.
The answer can-not be benzaldehyde because the potassium permanganate is a very strong reagent and it converts the benzaldehyde formed into benzoic acid. Firstly, from toluene benzaldehyde is formed which later gets converted into benzoic acid.
The reaction for this is as -

So, the answer is option a.)
Note: It must be noted that alkylation means the addition of alkyl groups. As here, the reagent was methyl chloride; this means the reagent had a methyl group. So, the methylation occurred. If it would have been ethyl chloride along with aluminium trichloride, the ethylation would have occurred.
Recently Updated Pages
JEE Main Mock Test 2025-26: Principles Related To Practical

JEE Main 2025-26 Mock Test: Organic Compounds Containing Nitrogen

JEE Main Mock Test 2025-26: Purification & Characterisation of Organic Compounds

JEE Main 2025-26 Mock Test: Principles Related To Practical

JEE Main Mock Test 2025-26: Principles & Best Practices

Purification and Characterisation of Organic Compounds JEE Main 2025-26 Mock Test

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

JEE Main Colleges 2026: Complete List of Participating Institutes

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Hybridisation in Chemistry – Concept, Types & Applications

Other Pages
JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 10 Biomolecules - 2025-26

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26




