In bleaching powder $(CaO{Cl}_{2})$, oxidation number of the two chlorine atom are:
(A) 0, +1
(B) 0, -1
(C) -1, +1
(D) -1, -2
Answer
263.7k+ views
Hint: In bleaching powder, $(CaO{Cl}_{2})$ the two chlorine atoms are attached to two different atoms and due to this they have different oxidation states. The oxidation number of an atom is defined as the charge that atom would have possessed if the compound was composed of ions.
Complete answer step by step: The oxidation number or oxidation state is the total no. of electrons that an atom either loses or gains in order to form chemical bonds with another atom. If the oxidation number is positive then that means the atom loses electrons and if it is negative it means that it gains the electrons.
Let us now look at bleaching powder. The chemical name of bleaching powder is calcium hypochlorite or calcium oxychloride and thus has a chemical formula as $CaO{Cl}_{2}$. It acts as a bleaching agent in aqueous solutions only. In bleaching powder, $(CaO{Cl}_{2})$, the two chlorine atoms are attached to two different atoms. One of the chlorine atoms is attached to calcium and the other chlorine atom is attached to oxygen. Thus, the chemical formula can also be written as $Ca(OCl)Cl$. The structure of the same is given below:
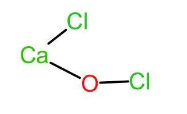
Let the oxidation state of Cl attached to calcium be x and oxidation state of Cl in $^{-}OCl$ be y.
We know that Ca is bivalent, therefore,
x = -1 and
2x + y = -1
Substituting the value of x, we get,
2(-1) + y = -1
$\implies $ y = +1
Therefore, the oxidation states of the two chlorine atoms in bleaching powder is -1 and +1.
Hence, option (c) is the correct answer.
Note: It is very important to note that oxidation numbers are always found out for individual atoms and not for groups of atoms. Therefore, in the above case, +1 is not the oxidation number of the group $^{-}OCl$ but of chlorine in $^{-}OCl$. The oxygen in $^{-}OCl$ has oxidation no. of -2.
Complete answer step by step: The oxidation number or oxidation state is the total no. of electrons that an atom either loses or gains in order to form chemical bonds with another atom. If the oxidation number is positive then that means the atom loses electrons and if it is negative it means that it gains the electrons.
Let us now look at bleaching powder. The chemical name of bleaching powder is calcium hypochlorite or calcium oxychloride and thus has a chemical formula as $CaO{Cl}_{2}$. It acts as a bleaching agent in aqueous solutions only. In bleaching powder, $(CaO{Cl}_{2})$, the two chlorine atoms are attached to two different atoms. One of the chlorine atoms is attached to calcium and the other chlorine atom is attached to oxygen. Thus, the chemical formula can also be written as $Ca(OCl)Cl$. The structure of the same is given below:

Let the oxidation state of Cl attached to calcium be x and oxidation state of Cl in $^{-}OCl$ be y.
We know that Ca is bivalent, therefore,
x = -1 and
2x + y = -1
Substituting the value of x, we get,
2(-1) + y = -1
$\implies $ y = +1
Therefore, the oxidation states of the two chlorine atoms in bleaching powder is -1 and +1.
Hence, option (c) is the correct answer.
Note: It is very important to note that oxidation numbers are always found out for individual atoms and not for groups of atoms. Therefore, in the above case, +1 is not the oxidation number of the group $^{-}OCl$ but of chlorine in $^{-}OCl$. The oxygen in $^{-}OCl$ has oxidation no. of -2.
Recently Updated Pages
JEE Main Mock Test 2025-26: Principles Related To Practical

JEE Main 2025-26 Experimental Skills Mock Test – Free Practice

JEE Main 2025-26 Electronic Devices Mock Test: Free Practice Online

JEE Main 2025-26 Mock Tests: Free Practice Papers & Solutions

JEE Main 2025-26: Magnetic Effects of Current & Magnetism Mock Test

JEE Main Statistics and Probability Mock Test 2025-26

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

NCERT Solutions For Class 11 Chemistry In Hindi Chapter 1 Some Basic Concepts Of Chemistry - 2025-26

How to Convert a Galvanometer into an Ammeter or Voltmeter

Understanding Electromagnetic Waves and Their Importance




