If \[A{B_4}\] molecule is a polar molecule, a possible geometry of \[A{B_4}\] is:
A. Square planar
B. Tetrahedral
C. Square pyramidal
D. Rectangular planar
Answer
249.3k+ views
Hint: When the atoms of a molecule are arranged in a three-dimensional structure it gives the molecular geometry of that molecule. Various geometrical parameters like bond length, torsional angles, molecular bond angles, etc., determine the shape of a molecule and the position of the atoms in it. The geometry of molecules is dependent on various factors like the presence or absence of lone pair in the central atom.
Complete Step by Step Solution:
A. If \[A{B_4}\] molecule is a square planar then it should be non-polar because the vector sum of the dipole moment here is zero.
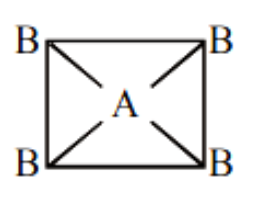
Image caption – Square planar geometry
B. If \[A{B_4}\] molecule is tetrahedral then it has no lone pair and their structure should be represented as follows:
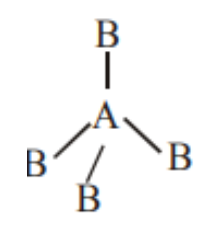
Image caption - Tetrahedral geometry
Here also it should be non-polar as it has perfect symmetry.
C. If \[A{B_4}\] molecule is a square pyramidal then it has one lone pair and their structure should be represented as:
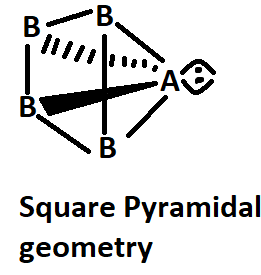
Image caption - Square pyramidal geometry
Here, in fact, the molecule is polar because the dipole moment of the lone pair of A is not affected by others.
(4) If \[A{B_4}\] molecule is a rectangular plane then it should be non-polar because the vector sum of the dipole moment is zero.
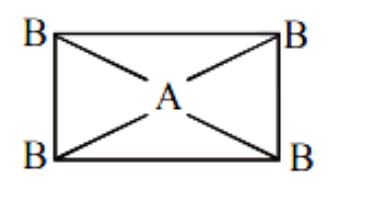
Image caption - Rectangular planar geometry
From the above analysis, it is evident that only the square pyramidal geometry satisfies the polar nature of the molecule. So it is our correct answer.
Hence, the correct answer is, option (C).
Note: It should be noted that the geometry of a molecule can be determined using the VSEPR model. The VSEPR model of organisation aids in making molecules more stable and less energetic.
Complete Step by Step Solution:
A. If \[A{B_4}\] molecule is a square planar then it should be non-polar because the vector sum of the dipole moment here is zero.

Image caption – Square planar geometry
B. If \[A{B_4}\] molecule is tetrahedral then it has no lone pair and their structure should be represented as follows:

Image caption - Tetrahedral geometry
Here also it should be non-polar as it has perfect symmetry.
C. If \[A{B_4}\] molecule is a square pyramidal then it has one lone pair and their structure should be represented as:

Image caption - Square pyramidal geometry
Here, in fact, the molecule is polar because the dipole moment of the lone pair of A is not affected by others.
(4) If \[A{B_4}\] molecule is a rectangular plane then it should be non-polar because the vector sum of the dipole moment is zero.

Image caption - Rectangular planar geometry
From the above analysis, it is evident that only the square pyramidal geometry satisfies the polar nature of the molecule. So it is our correct answer.
Hence, the correct answer is, option (C).
Note: It should be noted that the geometry of a molecule can be determined using the VSEPR model. The VSEPR model of organisation aids in making molecules more stable and less energetic.
Recently Updated Pages
JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

Isoelectronic Definition in Chemistry: Meaning, Examples & Trends

Ionisation Energy and Ionisation Potential Explained

Iodoform Reactions - Important Concepts and Tips for JEE

Introduction to Dimensions: Understanding the Basics

Instantaneous Velocity Explained: Formula, Examples & Graphs

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

Derivation of Equation of Trajectory Explained for Students

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Other Pages
JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

CBSE Notes Class 11 Chemistry Chapter 9 - Hydrocarbons - 2025-26

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

CBSE Notes Class 11 Chemistry Chapter 8 - Organic Chemistry Some Basic Principles And Techniques - 2025-26

JEE Advanced Weightage 2025 Chapter-Wise for Physics, Maths and Chemistry




