If a graph is plotted with the atomic number on x-axis and the square root of the frequency of a spectral line of characteristic X-rays on the y-axis, then it will be
A) A straight line passing through the origin
B) A parabola
C) A straight line not passing through the origin
D) A rectangular hyperbola
Answer
256.8k+ views
Hint: The atomic number or proton number is the number of protons found in the nucleus. A spectral line is a characteristic line in the entire continuous spectrum of an element, resulting from light emission and absorption in a narrow range of frequency when compared with the nearby frequencies.
Complete step by step solution:
The atomic number of an element can be related to the square root of the frequency of a spectral line of characteristic X-rays using Moseley’s law. Moseley’s law is an empirical law concerning the characteristic X-rays emitted by atoms. It states that the square root of the frequency of emitted X-rays is approximately proportional to the atomic number.
Mathematically, the law can be represented using the equation \[\sqrt{\upsilon }\propto Z\] where \[\upsilon \] is the frequency of the emitted X-ray.
The final equation of Moseley’s law can be written as \[\upsilon =A{(Z-b)}^2\] where and are constants depending upon the X-ray emission. Moseley measured and plotted the X-ray frequencies of about 40 elements of the periodic table following his law and observed the graph to be a straight line. The plot of the graph with atomic numbers on the x-axis and the square root of frequencies on the y-axis furnished a straight line not passing through the origin.
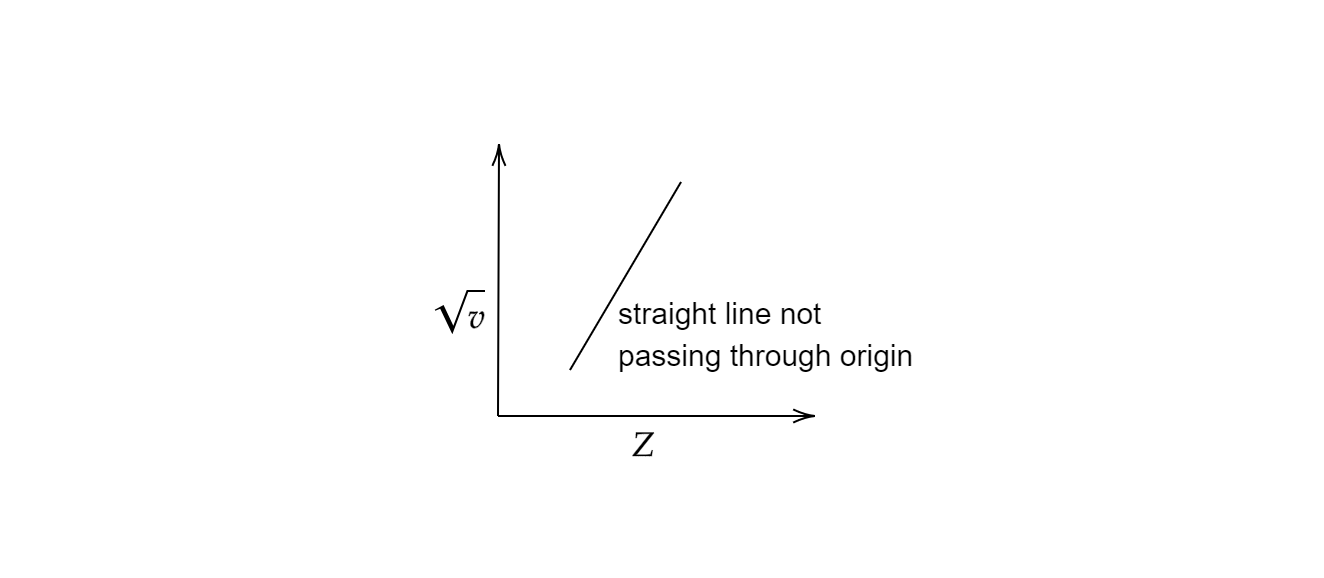
Hence, option (C) is the correct option.
Note: Moseley’s law was the first law to associate the atomic number of elements with any known physical quantity. It established the atomic number as a measurable experimental quantity and also gave it a viable physical meaning. Henry Moseley, the physicist behind this law, has also made major contributions to the periodic table.
Complete step by step solution:
The atomic number of an element can be related to the square root of the frequency of a spectral line of characteristic X-rays using Moseley’s law. Moseley’s law is an empirical law concerning the characteristic X-rays emitted by atoms. It states that the square root of the frequency of emitted X-rays is approximately proportional to the atomic number.
Mathematically, the law can be represented using the equation \[\sqrt{\upsilon }\propto Z\] where \[\upsilon \] is the frequency of the emitted X-ray.
The final equation of Moseley’s law can be written as \[\upsilon =A{(Z-b)}^2\] where and are constants depending upon the X-ray emission. Moseley measured and plotted the X-ray frequencies of about 40 elements of the periodic table following his law and observed the graph to be a straight line. The plot of the graph with atomic numbers on the x-axis and the square root of frequencies on the y-axis furnished a straight line not passing through the origin.

Hence, option (C) is the correct option.
Note: Moseley’s law was the first law to associate the atomic number of elements with any known physical quantity. It established the atomic number as a measurable experimental quantity and also gave it a viable physical meaning. Henry Moseley, the physicist behind this law, has also made major contributions to the periodic table.
Recently Updated Pages
Electricity and Magnetism Explained: Key Concepts & Applications

JEE Energetics Important Concepts and Tips for Exam Preparation

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

JEE Main Mock Test 2025-26: Electromagnetic Induction & Alternating Currents

JEE Main 2023 (February 1st Shift 1) Maths Question Paper with Answer Key

JEE Main 2023 (February 1st Shift 2) Maths Question Paper with Answer Key

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

JEE Main Colleges 2026: Complete List of Participating Institutes

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Hybridisation in Chemistry – Concept, Types & Applications

Other Pages
CBSE Class 12 Physics Question Paper 2026: Download SET-wise PDF with Answer Key & Analysis

JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

JEE Advanced Weightage 2025 Chapter-Wise for Physics, Maths and Chemistry

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students




