Identify ‘C’ in the following reaction:
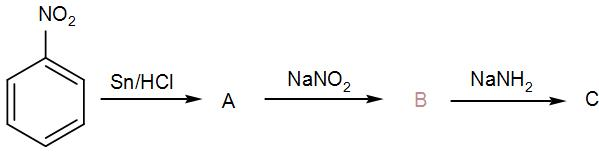
[A] Benzamide
[B] Benzoic acid
[C] Chlorobenzene
[D] Aniline
Answer
264.6k+ views
Hint: To solve this, proceed step wise and identify A, B and then arrive at C. Remember that tin with hydrochloric acid will reduce nitrobenzene and will give you an amine. Upon reaction with sodium nitride the amine will give you a salt. The salt will react with sodium amide and again arrive at phenyl amine.
Complete step by step solution:
To solve this, let’s see the reaction of nitrobenzene with the given reagents and then arrive at the final result.
In the first step, nitrobenzene reacts with tin and hydrochloric acid. It is a redox reaction and here the nitrobenzene is reduced to phenylamine. The reaction proceeds via formation of a phenyl ammonium cation by reduction of nitrobenzene. We can write the reaction as-
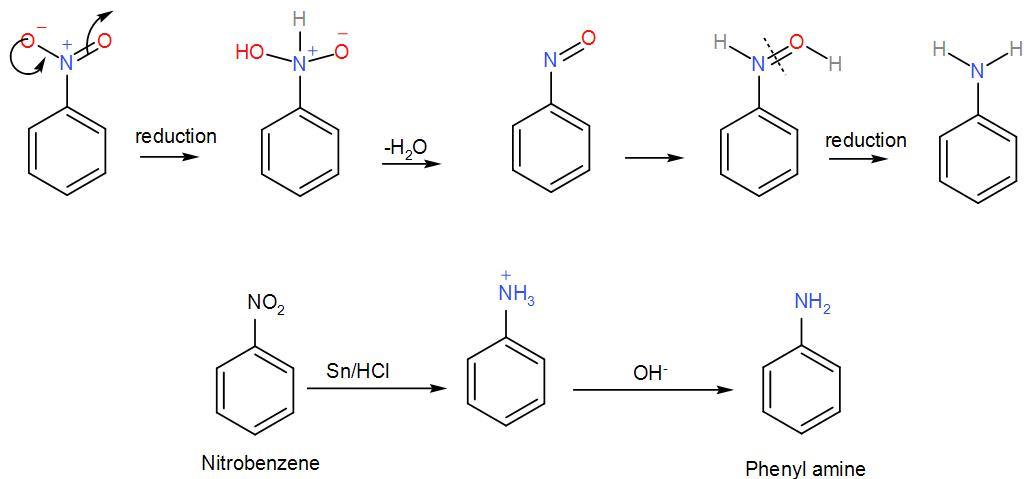
So, phenylamine is ‘A’. To this, sodium nitrite is added which will cause the amine to undergo diazotization. The hydrochloric acid in the reaction will react with it thus leading to the formation of a diazonium salt of benzene called benzene diazonium chloride. We can write the reaction as-
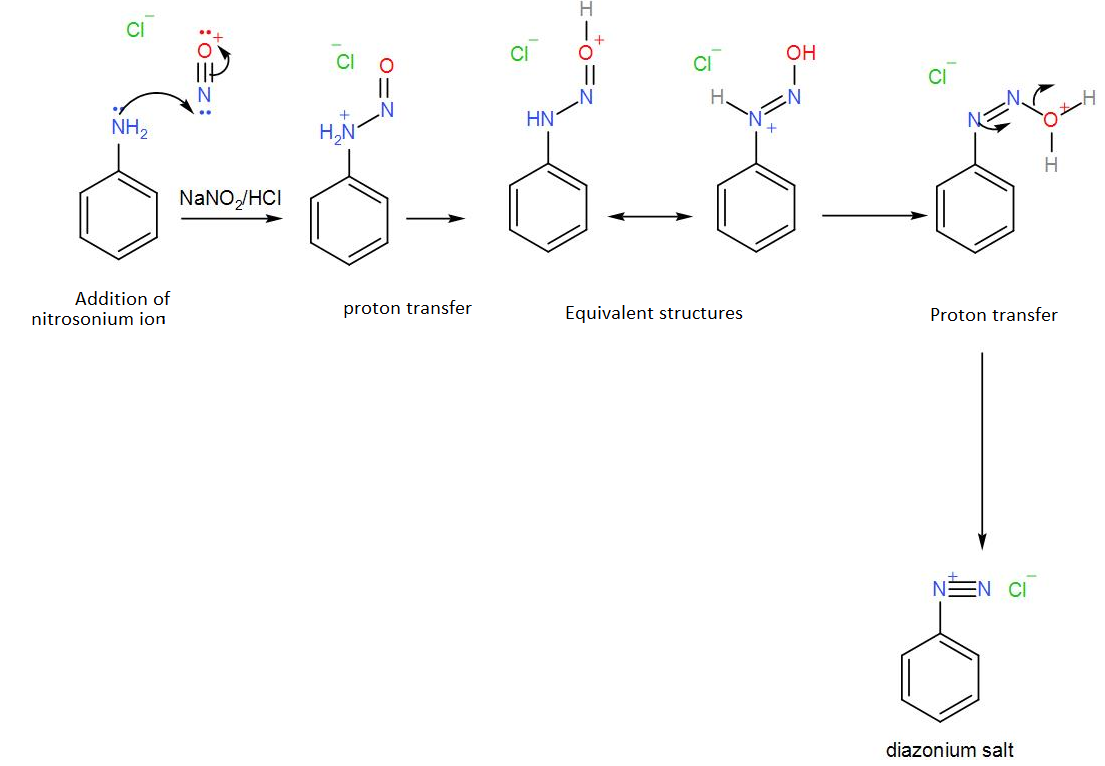
The diazonium salt thus obtained is our ‘B’.
To this diazonium salt when we add sodium amide we will again get the phenyl amine. We can write the reaction as-
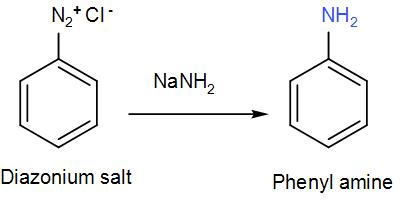
The phenyl amine is ‘C’. It is also known as aniline.
We can see from the above discussion that the ‘C’ in the reaction series is aniline.
Therefore, the correct answer is option [D] Aniline.
Note: Primary amines give us a diazonium salt which forms alcohols in addition to water but secondary amines have one hydrogen atom attached to them therefore, they cannot complete the diazotization reaction and give us a yellow oily nitrosamine product. Three degree amines have no hydrogen atoms attached to them thus they undergo simple acid- base reactions and give us soluble salts.
Complete step by step solution:
To solve this, let’s see the reaction of nitrobenzene with the given reagents and then arrive at the final result.
In the first step, nitrobenzene reacts with tin and hydrochloric acid. It is a redox reaction and here the nitrobenzene is reduced to phenylamine. The reaction proceeds via formation of a phenyl ammonium cation by reduction of nitrobenzene. We can write the reaction as-

So, phenylamine is ‘A’. To this, sodium nitrite is added which will cause the amine to undergo diazotization. The hydrochloric acid in the reaction will react with it thus leading to the formation of a diazonium salt of benzene called benzene diazonium chloride. We can write the reaction as-

The diazonium salt thus obtained is our ‘B’.
To this diazonium salt when we add sodium amide we will again get the phenyl amine. We can write the reaction as-

The phenyl amine is ‘C’. It is also known as aniline.
We can see from the above discussion that the ‘C’ in the reaction series is aniline.
Therefore, the correct answer is option [D] Aniline.
Note: Primary amines give us a diazonium salt which forms alcohols in addition to water but secondary amines have one hydrogen atom attached to them therefore, they cannot complete the diazotization reaction and give us a yellow oily nitrosamine product. Three degree amines have no hydrogen atoms attached to them thus they undergo simple acid- base reactions and give us soluble salts.
Recently Updated Pages
JEE Main Mock Test 2025-26: Principles Related To Practical

JEE Main 2025-26 Organic Compounds Containing Nitrogen Mock Test

JEE Main Chemical Kinetics Mock Test 2025-26: Free Practice Online

JEE Main 2025-26 Organic Compounds Containing Oxygen Mock Test

JEE Main 2025-26 Mock Test: Organic Compounds Containing Oxygen

JEE Main 2025-26 Organic Compounds Containing Halogens Mock Test

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

How to Convert a Galvanometer into an Ammeter or Voltmeter

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




