
Haloform reaction with ${{I}_{2}}$ and $KOH$ will be responded by:
A. 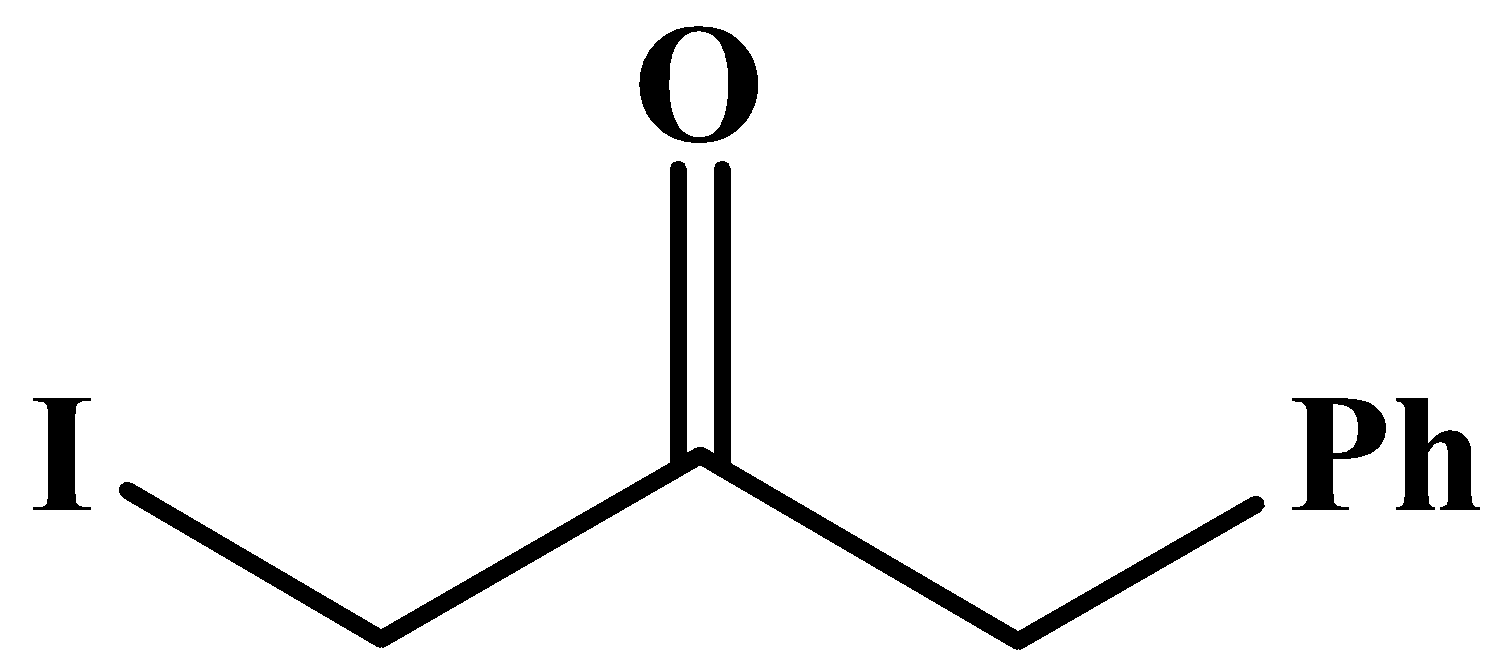
B. 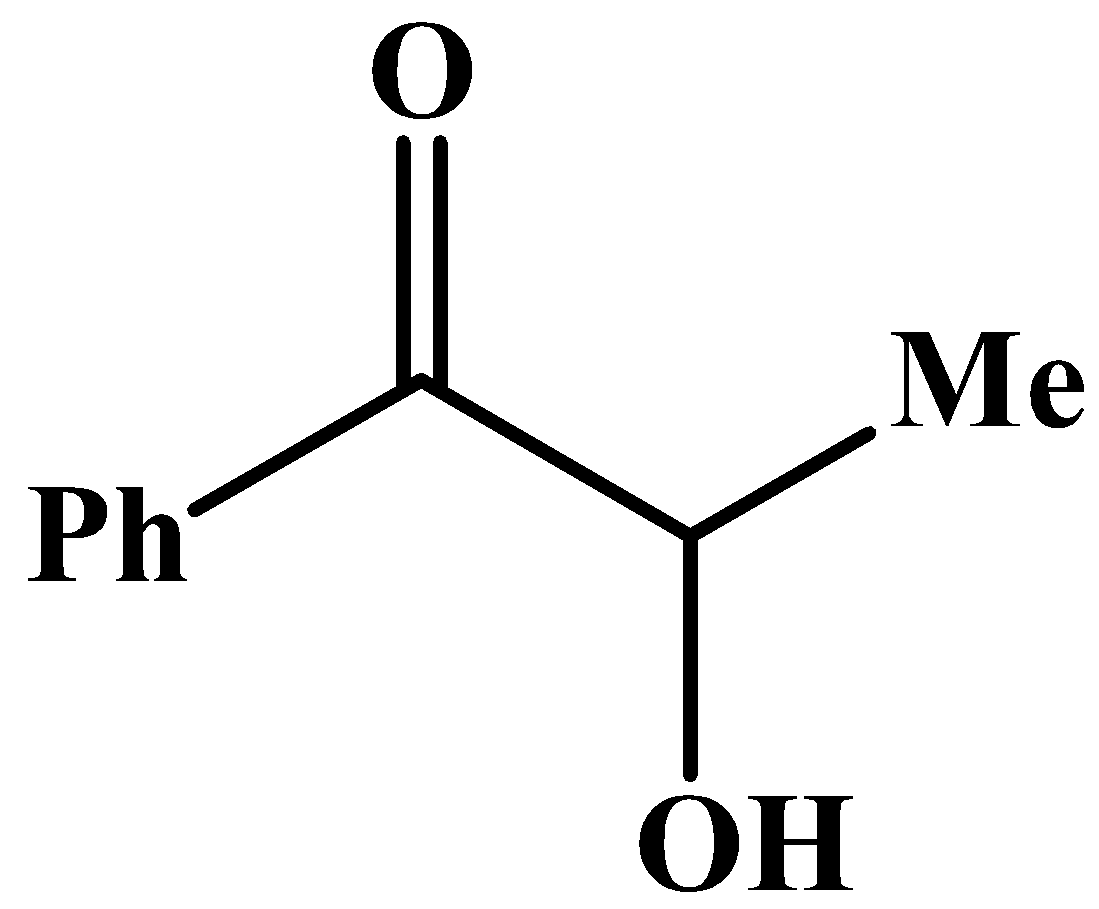
C. 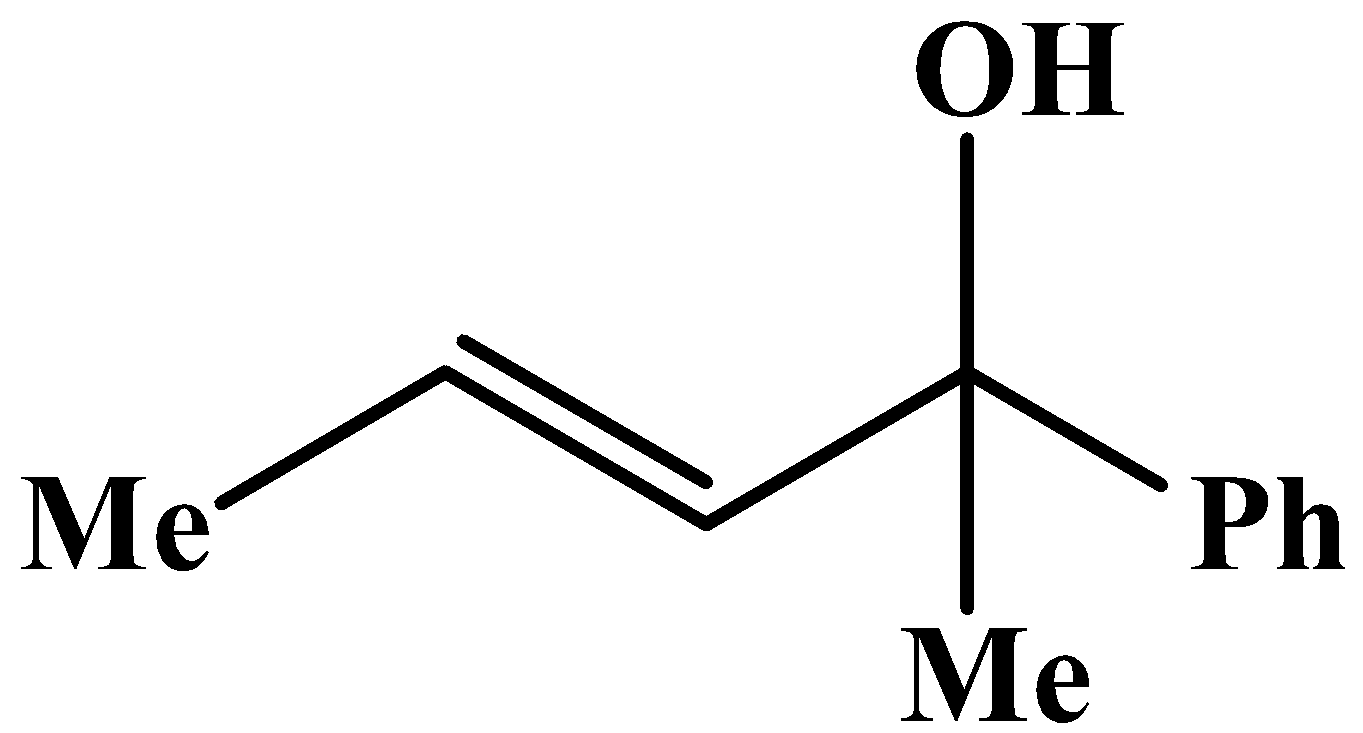
D. 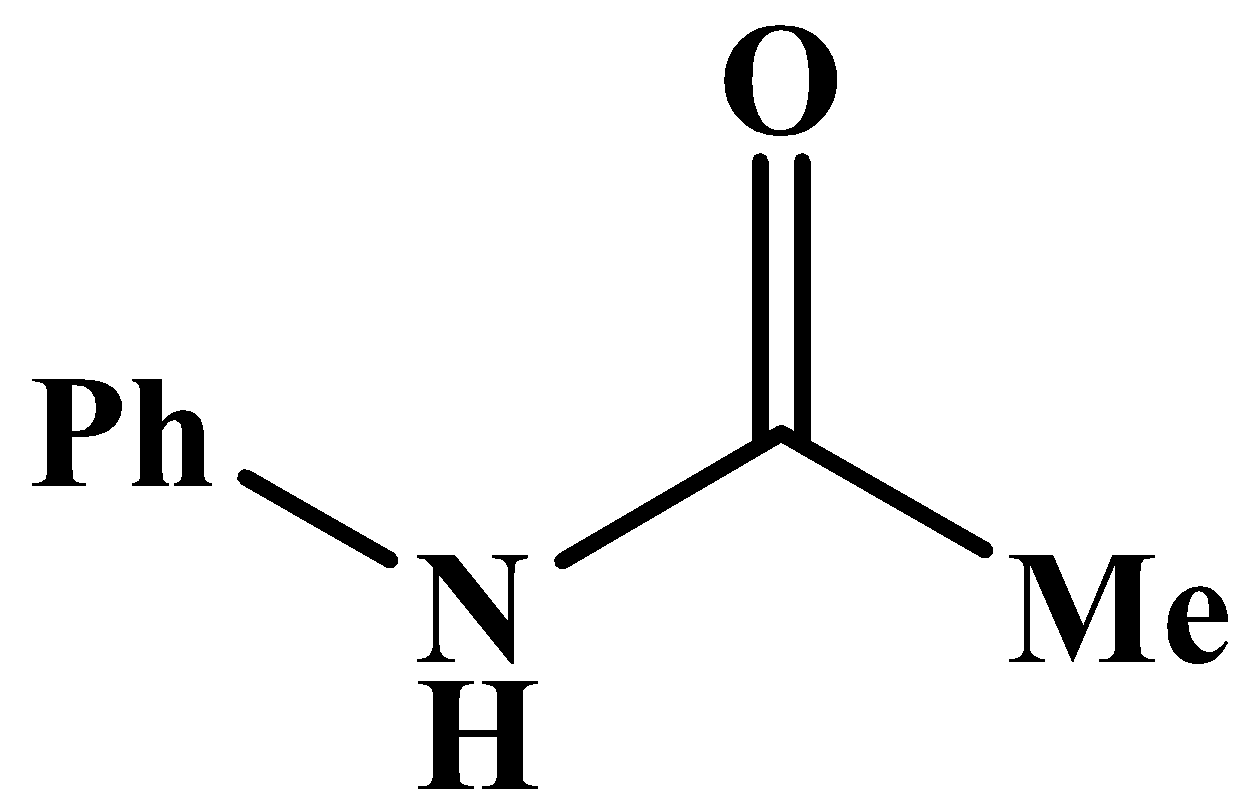
Answer
242.1k+ views
Hint: Haloform reaction is oxidising in nature. It requires a methyl group near a carbonyl carbon to give a positive test for this reaction.
Complete step-by-step answer:
The Haloform reaction is actually used to distinguish aldehydes and ketones which have a methyl group attached to the carbonyl carbon from the rest of the mixture. This reaction is oxidising in nature and therefore its products are a carboxylic acid which has one carbon atom less and a methyl halide. This is the same methyl group that was attached to the alpha carbon atom (the carbon atom bearing the carbonyl oxygen). The reaction is shown as below:
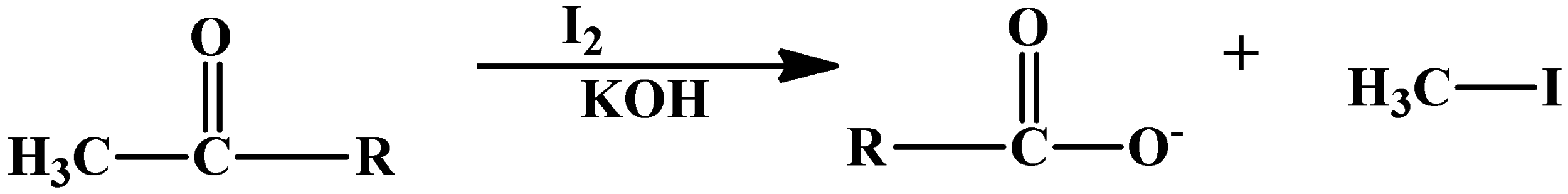
The “R” group here can be a hydrogen atom, making it an aldehyde or can be another alkyl group making it a ketone. This reaction does not affect other double bonds. So in a way the aromatic compounds can also undergo this process.
This reaction can also happen with alcohols, because they oxidise to carbonyl compounds. An example with ethyl alcohol is as below:
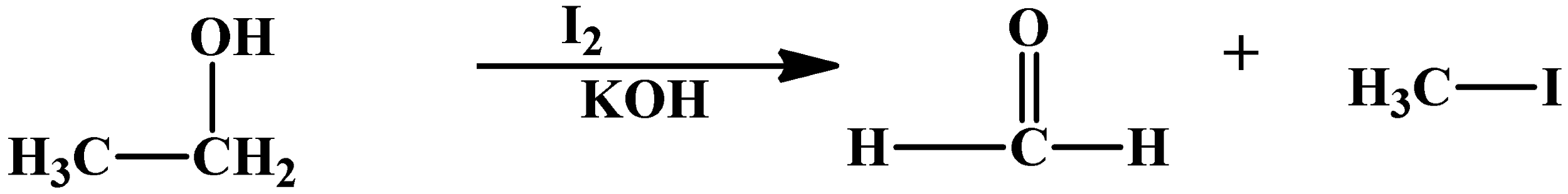
As you can see, ethyl alcohol loses a methyl group and then oxidises into formaldehyde. The by product is same as any Haloform reaction, which is a methyl halide.
Let’s look into the options that are given one-by-one:
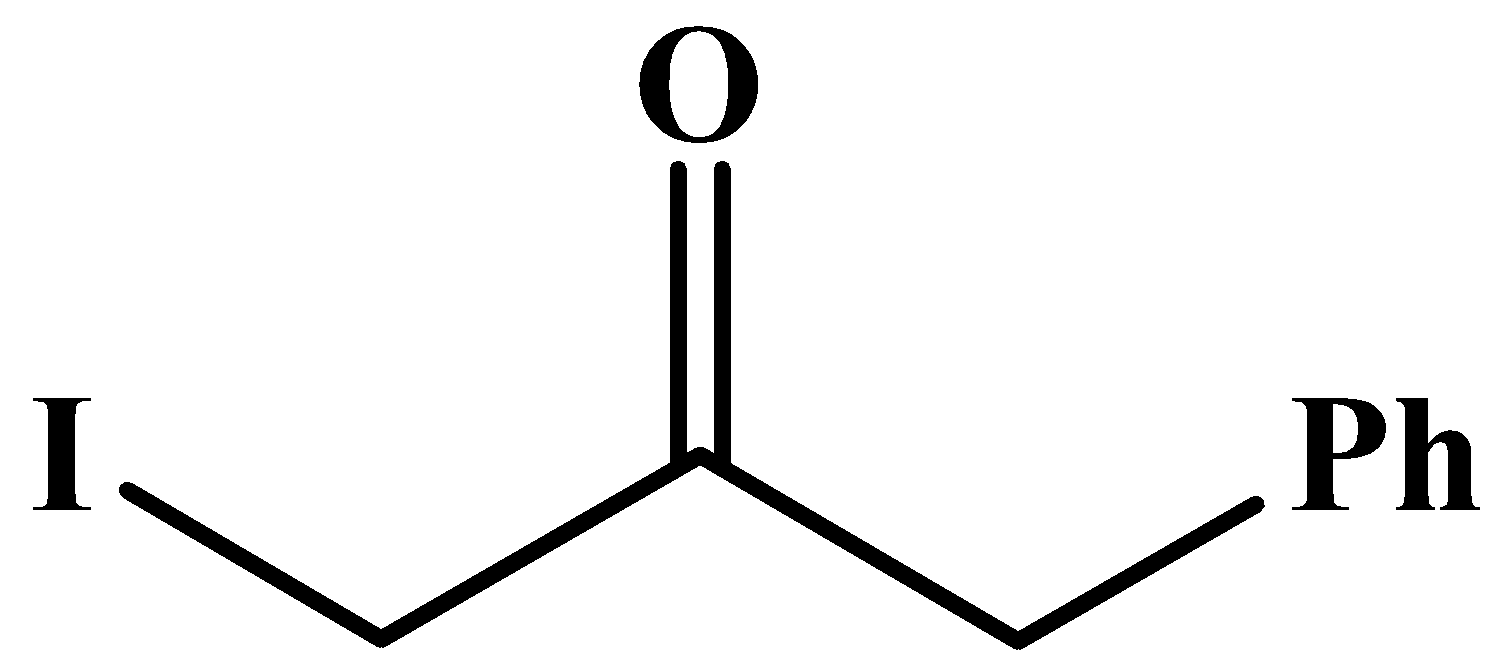
The carbonyl group here is in the middle of the compound and it has no methyl groups attached to it. Methyl groups are only possible at any end of a molecular chain or at the end of a branched chain.
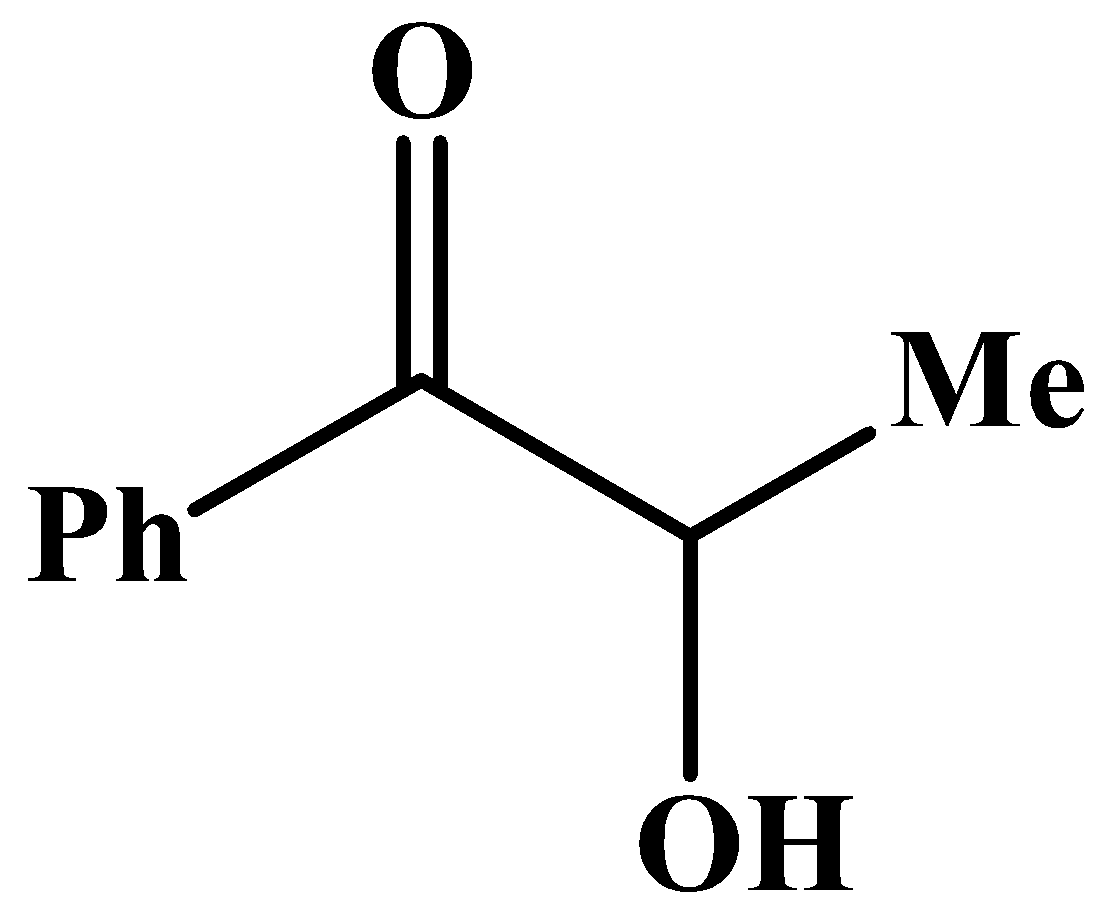
The carbonyl group has a phenyl group at its side and no other methyl group.
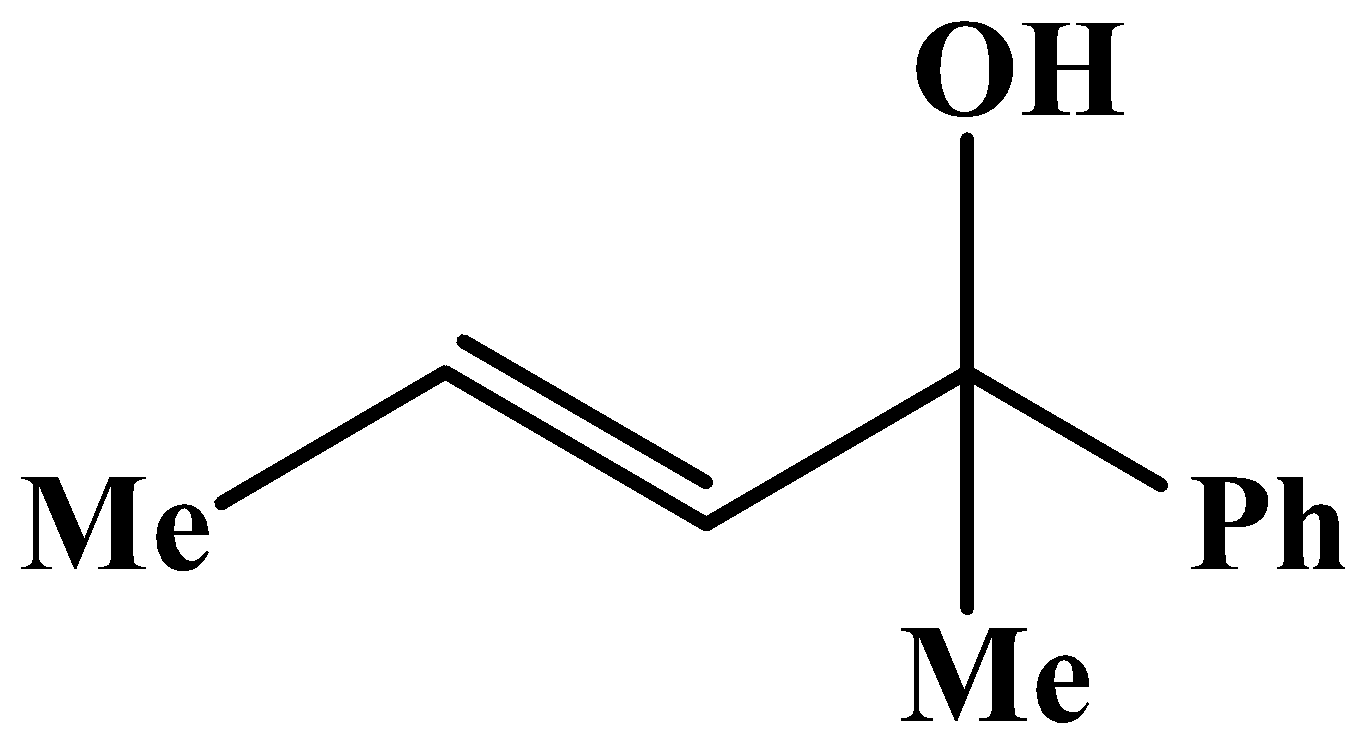
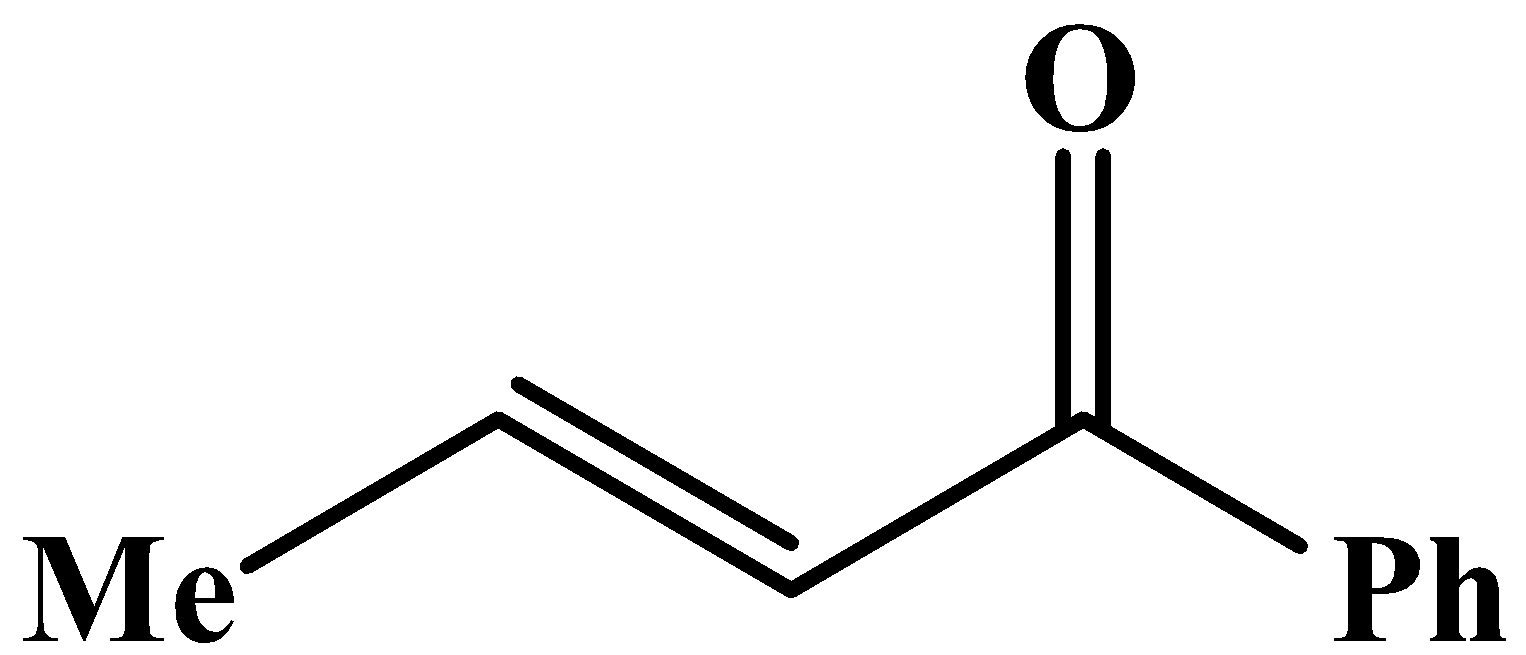
The methyl group is attached to the carbon bearing the hydroxyl group. This will respond to the oxidation reaction because it will convert into its corresponding carbonyl compound, which will be as shown below:
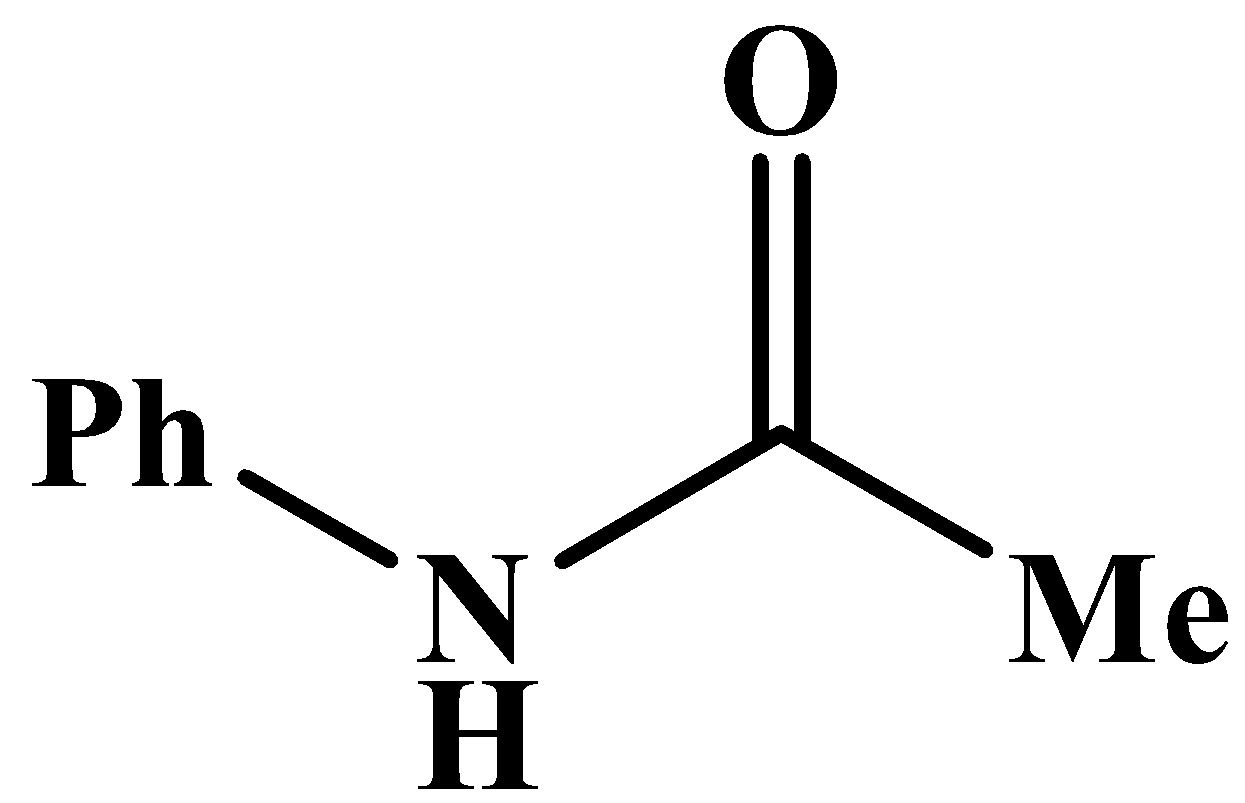
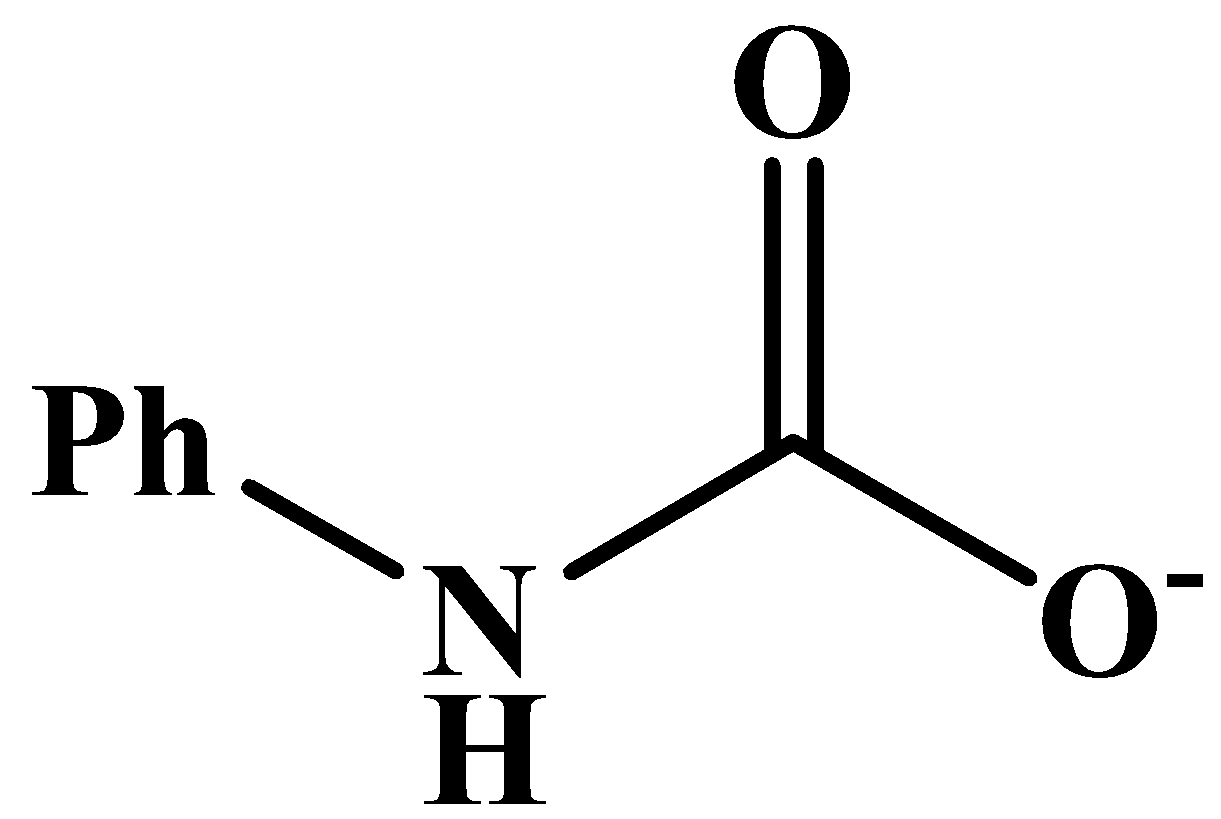
This will also respond to the given Haloform reaction as the methyl group is directly attached to the carbonyl carbon. The product of the reaction is:
Note:
The products of this reaction cannot be converted back into their original forms. This is because the compound that undergoes this reaction loses a methyl group, hence becoming shorter. Therefore this reaction is not used to oxidise carbonyl compounds into carboxylic acids or alcohols into aldehydes or ketones. This is only a reaction that can detect the presence of certain specific compounds. The by-products of this reaction are also a nuisance to separate.
Complete step-by-step answer:
The Haloform reaction is actually used to distinguish aldehydes and ketones which have a methyl group attached to the carbonyl carbon from the rest of the mixture. This reaction is oxidising in nature and therefore its products are a carboxylic acid which has one carbon atom less and a methyl halide. This is the same methyl group that was attached to the alpha carbon atom (the carbon atom bearing the carbonyl oxygen). The reaction is shown as below:

The “R” group here can be a hydrogen atom, making it an aldehyde or can be another alkyl group making it a ketone. This reaction does not affect other double bonds. So in a way the aromatic compounds can also undergo this process.
This reaction can also happen with alcohols, because they oxidise to carbonyl compounds. An example with ethyl alcohol is as below:

As you can see, ethyl alcohol loses a methyl group and then oxidises into formaldehyde. The by product is same as any Haloform reaction, which is a methyl halide.
Let’s look into the options that are given one-by-one:

The carbonyl group here is in the middle of the compound and it has no methyl groups attached to it. Methyl groups are only possible at any end of a molecular chain or at the end of a branched chain.

The carbonyl group has a phenyl group at its side and no other methyl group.

The methyl group is attached to the carbon bearing the hydroxyl group. This will respond to the oxidation reaction because it will convert into its corresponding carbonyl compound, which will be as shown below:


This will also respond to the given Haloform reaction as the methyl group is directly attached to the carbonyl carbon. The product of the reaction is:
Note:
The products of this reaction cannot be converted back into their original forms. This is because the compound that undergoes this reaction loses a methyl group, hence becoming shorter. Therefore this reaction is not used to oxidise carbonyl compounds into carboxylic acids or alcohols into aldehydes or ketones. This is only a reaction that can detect the presence of certain specific compounds. The by-products of this reaction are also a nuisance to separate.

Recently Updated Pages
Types of Solutions in Chemistry: Explained Simply

JEE Main Mock Test 2025-26: Principles Related To Practical

JEE Main 2025-26 Organic Compounds Containing Nitrogen Mock Test

JEE Main 2025-26 Mock Test: Organic Compounds Containing Oxygen

JEE Main 2025-26 Redox Reactions & Electro Mock Test

JEE Main Solutions Mock Test 1-2 (2025-26): Free Practice & Answers

Trending doubts
JEE Main 2026: Session 1 Results Out and Session 2 Registration Open, City Intimation Slip, Exam Dates, Syllabus & Eligibility

Ideal and Non-Ideal Solutions Explained for Class 12 Chemistry

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Clemmensen and Wolff Kishner Reductions Explained for JEE & NEET

Degree of Dissociation: Meaning, Formula, Calculation & Uses

Understanding the Angle of Deviation in a Prism

Other Pages
CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 10 Biomolecules - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 4 The D And F Block Elements - 2025-26

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More




