_________ grams of 3-Hydroxy propanal (MW=74) must be dehydrated to produce 7.8g of acrolein (MW=56) (\[{C_3}{H_4}O\]) if the percentage yield is 64. (Round off to the nearest integer).
[Given atomic masses; C=12.0 \[\mu \], H=1.0\[\mu \] , O=16.0\[\mu \]]
Answer
261.9k+ views
Hint: In the above-mentioned chemical reaction dehydration reaction is taking place. A dehydration reaction is a type of reaction in which there will be a loss of water from the chemical reaction. It is the reverse process of hydration reaction.
Complete Step by Step Solution:
When 3-Hydroxy propanal is dehydrated to produce 7.8g of acrolein, the chemical reaction is given as:
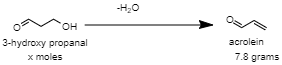
The moles of acrolein produced in the chemical reaction is:
\[\dfrac{{7.8}}{{56}} = 0.14\] moles
The percentage of yield would be equal to
\[\dfrac{{7.8/56}}{x} \times 100 = 64\]
\[x = \dfrac{{7.8 \times 100}}{{56 \times 64}} = \dfrac{{780}}{{56 \times 64}}\] moles
\[{W_{reac\tan t}} = \dfrac{{780}}{{56 \times 64}} \times 74 = 16.11\] grams
Therefore, 16.11 grams of 3-hydroxy propanal must be dehydrated to produce 7.8 grams of acrolein.
Note: Acrolein usually occurs as a colourless or yellow-colored liquid that dissolves in water very easily. It also has the tendency to quickly change into a vapour state when it is heated. Acrolein has a wide variety of uses such as in pesticides for controlling algae, weeds, bacteria, and mollusks. It also helps in the manufacturing of other industrial-based chemicals. It has a very choking odour and causes severe irritation to the eyes and mucous membrane. It is also toxic when it is inhaled and has a flash point which is below \[{0^o}F\]. It is less dense than water and its vapours are heavier than air. Very small amounts of acrolein can be produced and enter the atmosphere when oil. Plastics. Tobacco, gasoline, and oil are burned.
Complete Step by Step Solution:
When 3-Hydroxy propanal is dehydrated to produce 7.8g of acrolein, the chemical reaction is given as:

The moles of acrolein produced in the chemical reaction is:
\[\dfrac{{7.8}}{{56}} = 0.14\] moles
The percentage of yield would be equal to
\[\dfrac{{7.8/56}}{x} \times 100 = 64\]
\[x = \dfrac{{7.8 \times 100}}{{56 \times 64}} = \dfrac{{780}}{{56 \times 64}}\] moles
\[{W_{reac\tan t}} = \dfrac{{780}}{{56 \times 64}} \times 74 = 16.11\] grams
Therefore, 16.11 grams of 3-hydroxy propanal must be dehydrated to produce 7.8 grams of acrolein.
Note: Acrolein usually occurs as a colourless or yellow-colored liquid that dissolves in water very easily. It also has the tendency to quickly change into a vapour state when it is heated. Acrolein has a wide variety of uses such as in pesticides for controlling algae, weeds, bacteria, and mollusks. It also helps in the manufacturing of other industrial-based chemicals. It has a very choking odour and causes severe irritation to the eyes and mucous membrane. It is also toxic when it is inhaled and has a flash point which is below \[{0^o}F\]. It is less dense than water and its vapours are heavier than air. Very small amounts of acrolein can be produced and enter the atmosphere when oil. Plastics. Tobacco, gasoline, and oil are burned.
Recently Updated Pages
JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Electricity and Magnetism Explained: Key Concepts & Applications

JEE Energetics Important Concepts and Tips for Exam Preparation

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Understanding the Different Types of Solutions in Chemistry

Derivation of Equation of Trajectory Explained for Students

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

JEE Advanced Marks vs Rank 2025 - Predict Your IIT Rank Based on Score

Understanding Atomic Structure for Beginners




