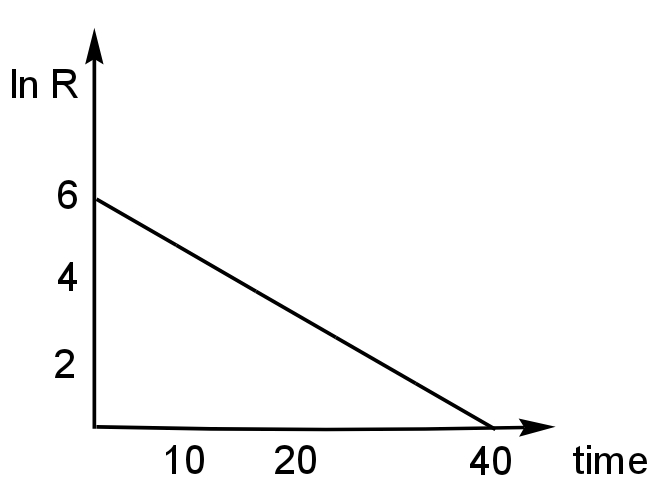
For the given graph between decay rate and time find half-life (where R = decay rate)
A. \[\dfrac{{10}}{3}\ln 2\]
B. \[\dfrac{{20}}{3}\ln 2\]
C. \[\dfrac{3}{{20}}\ln 2\]
D. \[\dfrac{3}{{10}}\ln 2\]
Answer
254.4k+ views
Hint: Radioactive reactions like the decaying of nuclei follow first-order kinetics. Here the rate constant is then depicted by the symbol λ. The rate of decay, or activity, of a species of radioactive material, is the reduction in the no.of radioactive nuclei per unit of time.
Formula Used:
\[{\rm{lnR = ln}}{{\rm{R}}_{\rm{o}}}{\rm{ - \lambda t}}\];
where, R= decay rate of a reactant at a time 't'.
\[{{\rm{R}}_{\rm{o}}}\]= initial decay rate of reactant
\[{\rm{\lambda }}{{\rm{t}}_{{\rm{1/2}}}}{\rm{ = ln2}}\]
\[{\rm{\lambda }}\]= rate constant
Complete Step by Step Solution:
The graph that is given in the question is a graph for a first-order reaction.
A reaction is called first-order if the rate of the reaction relies on one decay rate term only.
For the reaction:\[{\rm{A}} \to {\rm{B}}\]
,
the rate of reaction is directly proportional to the decay rate of A i.e., [A].
We know that for a first-order reaction,
\[{\rm{lnR = ln}}{{\rm{R}}_{\rm{o}}}{\rm{ - \lambda t}}\]-----(1)
The half-life of a reaction is the time in which the decay rate of a reactant is reduced to half of its initial decay rate.
It is depicted as .
So, \[{\rm{R = }}\dfrac{{{{\rm{R}}_{\rm{o}}}}}{{\rm{2}}}\]
Putting this value in the above-mentioned equation (1), we get
Hence,
\[{\rm{\lambda }}{{\rm{t}}_{{\rm{1/2}}}}{\rm{ = ln}}{{\rm{R}}_{\rm{o}}}{\rm{ - lnR}}\]
\[ \Rightarrow {\rm{\lambda }}{{\rm{t}}_{{\rm{1/2}}}}{\rm{ = }}\dfrac{{{\rm{ln}}{{\rm{R}}_{\rm{o}}}}}{{{\rm{ln}}\dfrac{{{{\rm{R}}_{\rm{o}}}}}{{\rm{2}}}}}\]
\[ \Rightarrow {\rm{\lambda }}{{\rm{t}}_{{\rm{1/2}}}}{\rm{ = ln2}}\]
Image: Graph plotted between lnR and time
Here, in the line and is plotted against time.
The equation for the first-order reaction, which is \[{\rm{lnR = ln}}{{\rm{R}}_{\rm{o}}}{\rm{ - \lambda t}}\] resembles the equation \[{\rm{y = mx + c}}\].
In this case,\[{\rm{y = lnR,x = t,m=- \lambda }}\].
So, slope, \[{\rm{m = - \lambda }}\]
Slope\[ = - \left( {\dfrac{6}{{40}}} \right) = - \left( {\dfrac{3}{{20}}} \right)\]
The slope is negative as this slope is decreasing.
\[{\rm{ - \lambda = }}\dfrac{{{\rm{ - 3}}}}{{20}}\]
So, \[{\rm{\lambda = }}\dfrac{{\rm{3}}}{{20}}\]
So, \[{{\rm{t}}_{{\rm{1/2}}}}{\rm{ = }}\dfrac{{{\rm{ln2}}}}{{\rm{\lambda }}}\]
=\[{\rm{ln}}\dfrac{{\rm{2}}}{{\left( {\dfrac{{\rm{3}}}{{{\rm{20}}}}} \right)}}\]
= \[\dfrac{3}{{20}}\ln 2\]
So, the half-life is \[\dfrac{3}{{20}}\ln 2\].
Note: While attempting the question, one must apply the appropriate form of rate law equation for a first-order reaction. He/she has to observe the values against which the given graph is plotted. Radioactive reactions work according to first-order kinetics. Radioactivity, or radioactive decay, is the emission of a particle or a photon that is due to the breakdown of the unstable nucleus of an atom.
Formula Used:
\[{\rm{lnR = ln}}{{\rm{R}}_{\rm{o}}}{\rm{ - \lambda t}}\];
where, R= decay rate of a reactant at a time 't'.
\[{{\rm{R}}_{\rm{o}}}\]= initial decay rate of reactant
\[{\rm{\lambda }}{{\rm{t}}_{{\rm{1/2}}}}{\rm{ = ln2}}\]
\[{\rm{\lambda }}\]= rate constant
Complete Step by Step Solution:
The graph that is given in the question is a graph for a first-order reaction.
A reaction is called first-order if the rate of the reaction relies on one decay rate term only.
For the reaction:\[{\rm{A}} \to {\rm{B}}\]
,
the rate of reaction is directly proportional to the decay rate of A i.e., [A].
We know that for a first-order reaction,
\[{\rm{lnR = ln}}{{\rm{R}}_{\rm{o}}}{\rm{ - \lambda t}}\]-----(1)
The half-life of a reaction is the time in which the decay rate of a reactant is reduced to half of its initial decay rate.
It is depicted as .
So, \[{\rm{R = }}\dfrac{{{{\rm{R}}_{\rm{o}}}}}{{\rm{2}}}\]
Putting this value in the above-mentioned equation (1), we get
Hence,
\[{\rm{\lambda }}{{\rm{t}}_{{\rm{1/2}}}}{\rm{ = ln}}{{\rm{R}}_{\rm{o}}}{\rm{ - lnR}}\]
\[ \Rightarrow {\rm{\lambda }}{{\rm{t}}_{{\rm{1/2}}}}{\rm{ = }}\dfrac{{{\rm{ln}}{{\rm{R}}_{\rm{o}}}}}{{{\rm{ln}}\dfrac{{{{\rm{R}}_{\rm{o}}}}}{{\rm{2}}}}}\]
\[ \Rightarrow {\rm{\lambda }}{{\rm{t}}_{{\rm{1/2}}}}{\rm{ = ln2}}\]
Image: Graph plotted between lnR and time
Here, in the line and is plotted against time.
The equation for the first-order reaction, which is \[{\rm{lnR = ln}}{{\rm{R}}_{\rm{o}}}{\rm{ - \lambda t}}\] resembles the equation \[{\rm{y = mx + c}}\].
In this case,\[{\rm{y = lnR,x = t,m=- \lambda }}\].
So, slope, \[{\rm{m = - \lambda }}\]
Slope\[ = - \left( {\dfrac{6}{{40}}} \right) = - \left( {\dfrac{3}{{20}}} \right)\]
The slope is negative as this slope is decreasing.
\[{\rm{ - \lambda = }}\dfrac{{{\rm{ - 3}}}}{{20}}\]
So, \[{\rm{\lambda = }}\dfrac{{\rm{3}}}{{20}}\]
So, \[{{\rm{t}}_{{\rm{1/2}}}}{\rm{ = }}\dfrac{{{\rm{ln2}}}}{{\rm{\lambda }}}\]
=\[{\rm{ln}}\dfrac{{\rm{2}}}{{\left( {\dfrac{{\rm{3}}}{{{\rm{20}}}}} \right)}}\]
= \[\dfrac{3}{{20}}\ln 2\]
So, the half-life is \[\dfrac{3}{{20}}\ln 2\].
Note: While attempting the question, one must apply the appropriate form of rate law equation for a first-order reaction. He/she has to observe the values against which the given graph is plotted. Radioactive reactions work according to first-order kinetics. Radioactivity, or radioactive decay, is the emission of a particle or a photon that is due to the breakdown of the unstable nucleus of an atom.
Recently Updated Pages
JEE Main 2022 (June 25th Shift 2) Chemistry Question Paper with Answer Key

Average Atomic Mass - Important Concepts and Tips for JEE

JEE Main 2023 (April 6th Shift 2) Chemistry Question Paper with Answer Key

JEE Main 2022 (June 27th Shift 2) Chemistry Question Paper with Answer Key

JEE Main 2023 (January 30th Shift 2) Maths Question Paper with Answer Key

JEE Main 2022 (July 29th Shift 1) Chemistry Question Paper with Answer Key

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Understanding the Different Types of Solutions in Chemistry

Other Pages
JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

CBSE Notes Class 11 Chemistry Chapter 9 - Hydrocarbons - 2025-26

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

CBSE Notes Class 11 Chemistry Chapter 8 - Organic Chemistry Some Basic Principles And Techniques - 2025-26

JEE Advanced Weightage 2025 Chapter-Wise for Physics, Maths and Chemistry




