Following Sidgwick’s rule of EAN, $\text{C}{{\text{o}}_{2}}{{\left( \text{CO} \right)}_{\text{x}}}$ will be:
A. $\text{C}{{\text{o}}_{2}}{{\left( \text{CO} \right)}_{4}}$
B. $\text{C}{{\text{o}}_{2}}{{\left( \text{CO} \right)}_{3}}$
C. $\text{C}{{\text{o}}_{2}}{{\left( \text{CO} \right)}_{8}}$
D. $\text{C}{{\text{o}}_{2}}{{\left( \text{CO} \right)}_{10}}$
Answer
268.5k+ views
Hint: EAN is the effective atomic number. The formula of EAN rule is Atomic number – oxidation state + electrons by ligands. Find the oxidation state, atomic number and electron donated. Solve the question with respect to one cobalt and get the value of x, this is because there is a bridging bond $\left( \text{Co}-\text{Co} \right)$ or symmetrical structure.
Complete step by step solution:
The EAN value of period 4 will be the atomic number of the last element present in this period. The last element of this period is Krypton $\left( \text{Kr} \right)$ with atomic number 36. The last element is taken because it is a noble gas and its configuration is noble gas configuration.
Let us solve this question step by step to find the final compound:
Step (1)- The EAN value of $\text{C}{{\text{o}}_{2}}{{\left( \text{CO} \right)}_{\text{x}}}$ will be 36.
Because cobalt also belongs to period 4.
Step (2)- The formula of EAN is the atomic number of central metal atom added to the number of electrons donated by ligands attached minus with its oxidation state. Mathematically, written as Atomic number – oxidation state + electrons by ligands.
We have done this question by considering only one cobalt and solving accordingly.
The atomic number of cobalt is 27.
The oxidation state of Co will be zero.
It is because the carbonyl compound has zero charge and overall charge on the compound is also.
The electrons donated by $\text{CO}$ ligand will be $\left( 2\times \text{no}\text{. of ligands} \right)$.
The EAN of $\text{C}{{\text{o}}_{2}}{{\left( \text{CO} \right)}_{\text{x}}}$ will be $\left[ 27-0+1+\dfrac{\left( \text{2}\times \text{x} \right)}{2} \right]=36$.
Step (3)- Find the value of x.
The value of x will be $\left[ 28+\text{x} \right]=36$ or x = 8.
The compound formed will be $\text{C}{{\text{o}}_{2}}{{\left( \text{CO} \right)}_{8}}$. The IUPAC name of the compound is dicobalt octacarbonyl.
Following Sidgwick’s rule of EAN, $\text{C}{{\text{o}}_{2}}{{\left( \text{CO} \right)}_{\text{x}}}$ will be $\text{C}{{\text{o}}_{2}}{{\left( \text{CO} \right)}_{8}}$.
The correct option is option (C).
Additional Information:
Dicobalt octacarbonyl compound or $\text{C}{{\text{o}}_{2}}{{\left( \text{CO} \right)}_{8}}$ compound is used as a reagent and catalyst in organic synthesis and organometallic chemistry.
Note: Each molecule consists of two cobalt atoms and eight carbon monoxide ligands. The structure of $\text{C}{{\text{o}}_{2}}{{\left( \text{CO} \right)}_{8}}$ is
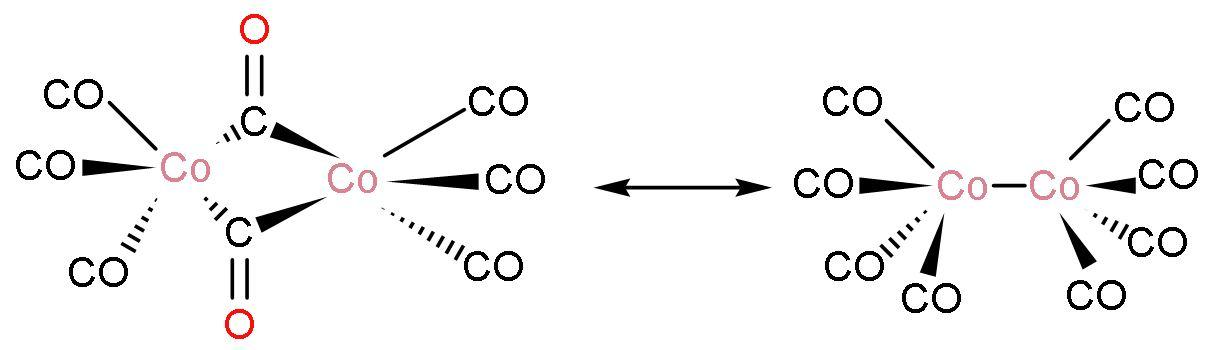
The compound exists in several isomeric forms. In solution, there are two isomers known that rapidly interconvert. In one structure, there is $\left( \text{Co}-\text{Co} \right)$ and eight $\left( \text{Co}-\text{CO} \right)$ ligands. Other structure has there is two bridging $\left( \text{Co}-\text{CO}-\text{Co} \right)$ ligand and there is six $\left( \text{Co}-\text{CO} \right)$ ligands.
Complete step by step solution:
The EAN value of period 4 will be the atomic number of the last element present in this period. The last element of this period is Krypton $\left( \text{Kr} \right)$ with atomic number 36. The last element is taken because it is a noble gas and its configuration is noble gas configuration.
Let us solve this question step by step to find the final compound:
Step (1)- The EAN value of $\text{C}{{\text{o}}_{2}}{{\left( \text{CO} \right)}_{\text{x}}}$ will be 36.
Because cobalt also belongs to period 4.
Step (2)- The formula of EAN is the atomic number of central metal atom added to the number of electrons donated by ligands attached minus with its oxidation state. Mathematically, written as Atomic number – oxidation state + electrons by ligands.
We have done this question by considering only one cobalt and solving accordingly.
The atomic number of cobalt is 27.
The oxidation state of Co will be zero.
It is because the carbonyl compound has zero charge and overall charge on the compound is also.
The electrons donated by $\text{CO}$ ligand will be $\left( 2\times \text{no}\text{. of ligands} \right)$.
The EAN of $\text{C}{{\text{o}}_{2}}{{\left( \text{CO} \right)}_{\text{x}}}$ will be $\left[ 27-0+1+\dfrac{\left( \text{2}\times \text{x} \right)}{2} \right]=36$.
Step (3)- Find the value of x.
The value of x will be $\left[ 28+\text{x} \right]=36$ or x = 8.
The compound formed will be $\text{C}{{\text{o}}_{2}}{{\left( \text{CO} \right)}_{8}}$. The IUPAC name of the compound is dicobalt octacarbonyl.
Following Sidgwick’s rule of EAN, $\text{C}{{\text{o}}_{2}}{{\left( \text{CO} \right)}_{\text{x}}}$ will be $\text{C}{{\text{o}}_{2}}{{\left( \text{CO} \right)}_{8}}$.
The correct option is option (C).
Additional Information:
Dicobalt octacarbonyl compound or $\text{C}{{\text{o}}_{2}}{{\left( \text{CO} \right)}_{8}}$ compound is used as a reagent and catalyst in organic synthesis and organometallic chemistry.
Note: Each molecule consists of two cobalt atoms and eight carbon monoxide ligands. The structure of $\text{C}{{\text{o}}_{2}}{{\left( \text{CO} \right)}_{8}}$ is

The compound exists in several isomeric forms. In solution, there are two isomers known that rapidly interconvert. In one structure, there is $\left( \text{Co}-\text{Co} \right)$ and eight $\left( \text{Co}-\text{CO} \right)$ ligands. Other structure has there is two bridging $\left( \text{Co}-\text{CO}-\text{Co} \right)$ ligand and there is six $\left( \text{Co}-\text{CO} \right)$ ligands.
Recently Updated Pages
Algebra Made Easy: Step-by-Step Guide for Students

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

JEE Energetics Important Concepts and Tips for Exam Preparation

Chemical Properties of Hydrogen - Important Concepts for JEE Exam Preparation

JEE General Topics in Chemistry Important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

JEE Advanced 2026 Marks vs Rank: Estimate IIT Rank from Your Score

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

Understanding the Different Types of Solutions in Chemistry

NCERT Solutions For Class 11 Chemistry In Hindi Chapter 1 Some Basic Concepts Of Chemistry - 2025-26




