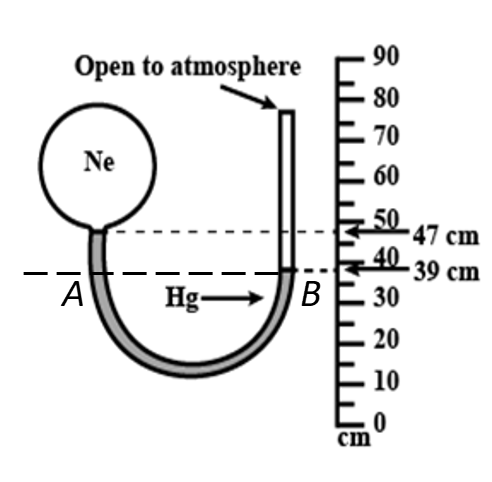
Find neon pressure in the manometer, when atmospheric pressure is 650 torr.
A) 665 torr
B) 80 torr
C) 570 torr
D) 650 torr
Answer
258.3k+ views
Hint: Manometer is a device that works on the principle of hydrostatic equilibrium. Hydrostatic equilibrium states that the pressure at any point in a fluid at rest is equal, and its value is just the weight of the overlying liquid.
Complete step by step answer:
Moving back to the question figure, we can see that height h of the manometric fluid column above point A is,
$h = 49\;{\rm{cm}} - 39\;{\rm{cm}}$
$ \Rightarrow h = 8\;{\rm{cm}} \times \dfrac{{10\;{\rm{mm}}}}{{1\;{\rm{cm}}}}$
$ \Rightarrow h = 80\;{\rm{mm}}$
Since point A and B are at the same levels, therefore the pressure at point A and B is the same, and it can be written as,
$ \Rightarrow {P_A} = {P_B}$
Now, the pressure at point A can also be written as the sum of the pressure of neon gas and the pressure to manometric fluid column h. Therefore,
$ \Rightarrow {P_{Ne}} + h = {P_A}$ …… (I)
Here ${P_{Ne}}$ is the pressure due to neon gas.
But since ${P_A} = {P_B}$ therefore the equation (I) becomes,
$ \Rightarrow {P_{Ne}} + h = {P_B}$ …… (II)
Since point B is open to atmosphere and therefore, the pressure at point B is equal to the atmospheric pressure, i.e. ${P_B} = 650\;{\rm{torr}}$.
We will now substitute ${P_B} = 650\;{\rm{torr}}$ and $h = 80\;{\rm{mm}}$ in equation (II), and therefore it becomes,
$ \Rightarrow {P_{Ne}} + 80\;{\rm{mm}} = 650\;{\rm{torr}} \times \dfrac{{1\;{\rm{mm}}}}{{1\;{\rm{torr}}}}$
$ \Rightarrow {P_{Ne}} + 80\;{\rm{mm}} = 650\;{\rm{mm}}$
$ \Rightarrow {P_{Ne}} = 570\;{\rm{mm}} \times \dfrac{{1\;{\rm{torr}}}}{{1\;{\rm{mm}}}}$
$ \Rightarrow {P_{Ne}} = 570\;{\rm{torr}}$
Therefore, the neon pressure in the manometer is 570 torr, and the correct option is (C).
Note: Since we have seen how a manometer is used, let us know some more applications of a manometer in daily life. In addition to straight pressure and vacuum measurement, other process variables that are a function of pressure can be readily measured with a manometer. Typical uses are flow, filter pressure drop, meter calibrations, leak testing and tank liquid level.
Complete step by step answer:
Moving back to the question figure, we can see that height h of the manometric fluid column above point A is,
$h = 49\;{\rm{cm}} - 39\;{\rm{cm}}$
$ \Rightarrow h = 8\;{\rm{cm}} \times \dfrac{{10\;{\rm{mm}}}}{{1\;{\rm{cm}}}}$
$ \Rightarrow h = 80\;{\rm{mm}}$
Since point A and B are at the same levels, therefore the pressure at point A and B is the same, and it can be written as,
$ \Rightarrow {P_A} = {P_B}$
Now, the pressure at point A can also be written as the sum of the pressure of neon gas and the pressure to manometric fluid column h. Therefore,
$ \Rightarrow {P_{Ne}} + h = {P_A}$ …… (I)
Here ${P_{Ne}}$ is the pressure due to neon gas.
But since ${P_A} = {P_B}$ therefore the equation (I) becomes,
$ \Rightarrow {P_{Ne}} + h = {P_B}$ …… (II)
Since point B is open to atmosphere and therefore, the pressure at point B is equal to the atmospheric pressure, i.e. ${P_B} = 650\;{\rm{torr}}$.
We will now substitute ${P_B} = 650\;{\rm{torr}}$ and $h = 80\;{\rm{mm}}$ in equation (II), and therefore it becomes,
$ \Rightarrow {P_{Ne}} + 80\;{\rm{mm}} = 650\;{\rm{torr}} \times \dfrac{{1\;{\rm{mm}}}}{{1\;{\rm{torr}}}}$
$ \Rightarrow {P_{Ne}} + 80\;{\rm{mm}} = 650\;{\rm{mm}}$
$ \Rightarrow {P_{Ne}} = 570\;{\rm{mm}} \times \dfrac{{1\;{\rm{torr}}}}{{1\;{\rm{mm}}}}$
$ \Rightarrow {P_{Ne}} = 570\;{\rm{torr}}$
Therefore, the neon pressure in the manometer is 570 torr, and the correct option is (C).
Note: Since we have seen how a manometer is used, let us know some more applications of a manometer in daily life. In addition to straight pressure and vacuum measurement, other process variables that are a function of pressure can be readily measured with a manometer. Typical uses are flow, filter pressure drop, meter calibrations, leak testing and tank liquid level.
Recently Updated Pages
Circuit Switching vs Packet Switching: Key Differences Explained

Dimensions of Pressure in Physics: Formula, Derivation & SI Unit

JEE General Topics in Chemistry Important Concepts and Tips

JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

JEE Main Colleges 2026: Complete List of Participating Institutes

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Hybridisation in Chemistry – Concept, Types & Applications

Other Pages
JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

CBSE Notes Class 11 Physics Chapter 1 - Units And Measurements - 2025-26

NCERT Solutions For Class 11 Physics Chapter 1 Units And Measurements - 2025-26

Important Questions For Class 11 Physics Chapter 1 Units and Measurement - 2025-26

JEE Advanced Weightage 2025 Chapter-Wise for Physics, Maths and Chemistry




