Explain the following name reaction with a suitable chemical reaction.
Cannizaro’s reaction.
Answer
269.4k+ views
Hint: This reaction is named after its discoverer Stanislao Cannizzaro. You should know that the products formed from this reaction are primary alcohol and a carboxylic acid. Now try solving the question.
Complete step by step answer:
> Let us know this reaction in detail.
The Cannizzaro reaction is a redox reaction in which two molecules of an aldehyde are reacted to produce a primary alcohol and a carboxylic acid using a hydroxide base.
> Let us see the mechanism of Cannizzaro reaction.
- You should know that the reaction begins with hydroxide attack on the carbonyl carbon followed by deprotonation to give a dianion. This unstable intermediate releases a hydride anion which attacks another molecule of aldehyde.
- In this process the dianion converts to a carboxylate anion and the aldehyde to an alkoxide.
The alkoxide in the reaction then picks up a proton from water to provide the alcohol as the final product, while the carboxylate is converted to the carboxylic acid product after acid work-up.
Let us understand better with the help of the reaction.
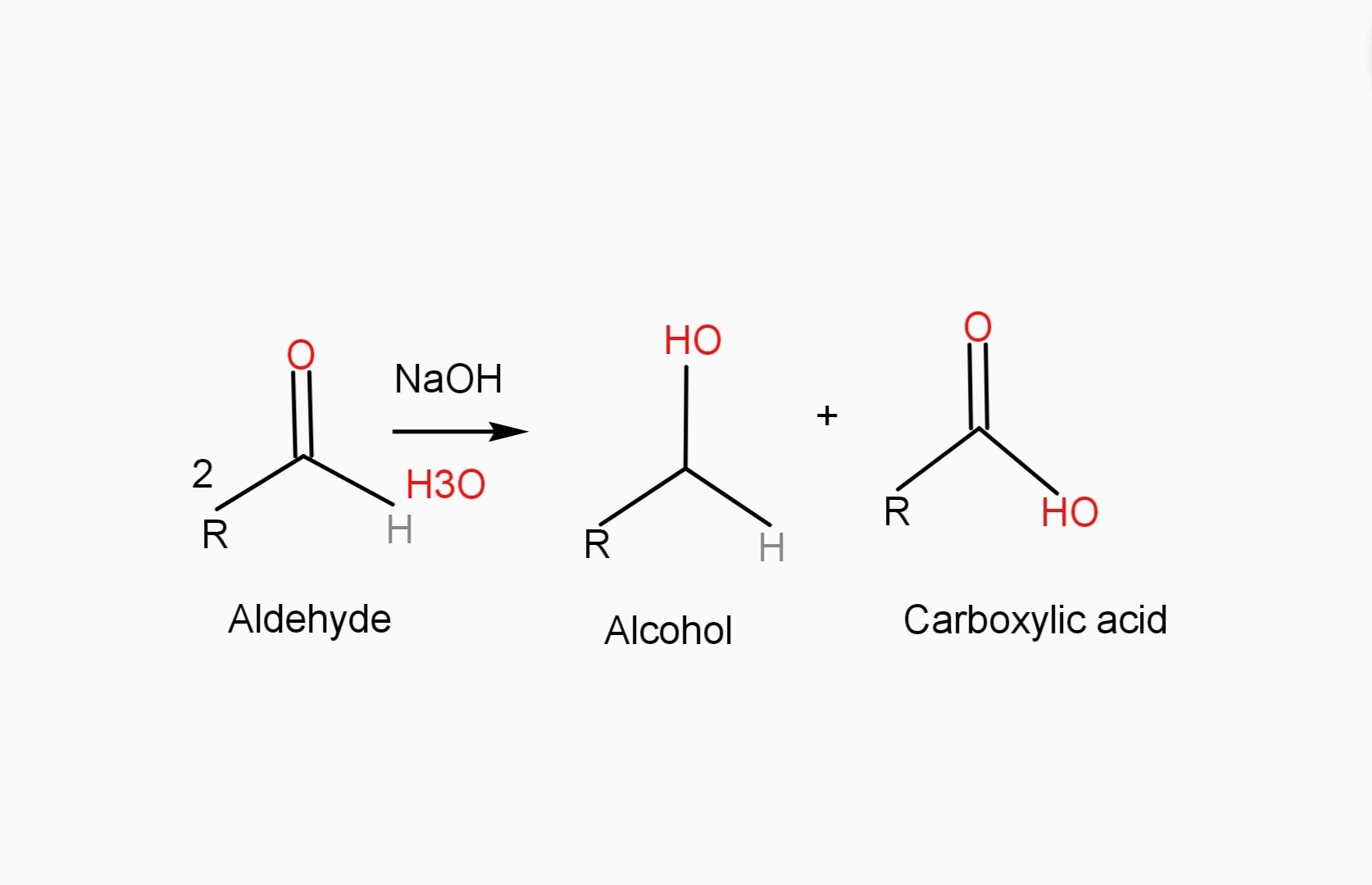
Example of Cannizzaro reaction , when formaldehyde is disproportionated to formic acid and methyl alcohol in strong alkali.
$HCHO\xrightarrow [ NaOH ]{ } HCO{ O }^{ - }{ Na }^{ + }+{ CH }_{ 3 }OH$
Note: The variations of the reaction improve the yield of the desired product. The Cannizzaro reaction can be used to influence a disproportionation of two molecules of a non-enolizable aldehyde to give a primary alcohol and a carboxylic acid.
Complete step by step answer:
> Let us know this reaction in detail.
The Cannizzaro reaction is a redox reaction in which two molecules of an aldehyde are reacted to produce a primary alcohol and a carboxylic acid using a hydroxide base.
> Let us see the mechanism of Cannizzaro reaction.
- You should know that the reaction begins with hydroxide attack on the carbonyl carbon followed by deprotonation to give a dianion. This unstable intermediate releases a hydride anion which attacks another molecule of aldehyde.
- In this process the dianion converts to a carboxylate anion and the aldehyde to an alkoxide.
The alkoxide in the reaction then picks up a proton from water to provide the alcohol as the final product, while the carboxylate is converted to the carboxylic acid product after acid work-up.
Let us understand better with the help of the reaction.

Example of Cannizzaro reaction , when formaldehyde is disproportionated to formic acid and methyl alcohol in strong alkali.
$HCHO\xrightarrow [ NaOH ]{ } HCO{ O }^{ - }{ Na }^{ + }+{ CH }_{ 3 }OH$
Note: The variations of the reaction improve the yield of the desired product. The Cannizzaro reaction can be used to influence a disproportionation of two molecules of a non-enolizable aldehyde to give a primary alcohol and a carboxylic acid.
Recently Updated Pages
JEE General Topics in Chemistry Important Concepts and Tips

JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Electricity and Magnetism Explained: Key Concepts & Applications

JEE Energetics Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




