What is the electronic configuration for a gas phase +3 ion of iron (Z = 26)?
(A) $\left[ Ar \right]3{{d}^{5}}$
(B) $\left[ Ar \right]4{{s}^{2}}3{{d}^{3}}$
(C) $\left[ Ar \right]4{{s}^{1}}3{{d}^{4}}$
(D) $\left[ Ar \right]4{{s}^{2}}3{{d}^{6}}$
Answer
249k+ views
Hint: Use the atomic number of iron given to write the electronic configuration of iron. A positive charge means electrons are deficient and a negative charge means electrons are in excess. Here it has a +3 positive charge therefore there we have to consider 3 fewer electrons while writing the configuration.
Complete step by step solution:
Electronic configuration of an atom or molecule gives us the numeric arrangement of electrons around the nucleus.
There are specific notations that we use for writing the configuration of an atom. For writing these notations, we start with the energy orbitals. Practically, there are 4 orbitals s, p, d and f. There is a certain even number of the electrons that each orbital can accommodate. s-orbital can accommodate 2 electrons whereas p, d and f-orbitals can accommodate 6, 10 and 14 electrons respectively.
There is a trend each electron follows while filling these orbitals and it is given as-
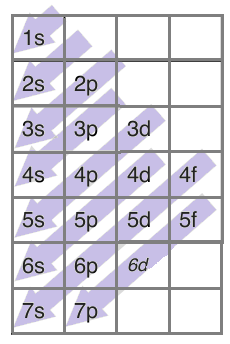
Following this trend, we can write the electronic configuration of the +3 state of iron.
We know that the atomic number of iron is 26. In +3 state it will lose 3 electrons therefore, we will be left with 23electrons. Following the above trend, we can write the electronic configuration of $F{{e}^{3+}}$ as- $1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}3{{s}^{2}}3{{p}^{6}}3{{d}^{5}}$
We know that the atomic number of argon is 18. Therefore, electronic configuration of argon is $1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}3{{s}^{2}}3{{p}^{6}}$ .
As we know argon lies in the third period in the periodic table and iron lies in the fourth period. Therefore, we can write the electronic configuration of iron in terms of the argon as- $\left[ Ar \right]3{{d}^{5}}$ .
Therefore, the correct answer is option (A) $\left[ Ar \right]3{{d}^{5}}$.
Note: According to Pauli’s Exclusion Principle, each orbital can hold 2 electrons. s-orbital set contains one orbital, thus can hold 2 electrons. Similarly, the p-orbital set had three orbitals, thus can hold 6 electrons and d-orbital and f-orbital have five and seven orbitals thus, can hold 10 and 14 electrons respectively.
Complete step by step solution:
Electronic configuration of an atom or molecule gives us the numeric arrangement of electrons around the nucleus.
There are specific notations that we use for writing the configuration of an atom. For writing these notations, we start with the energy orbitals. Practically, there are 4 orbitals s, p, d and f. There is a certain even number of the electrons that each orbital can accommodate. s-orbital can accommodate 2 electrons whereas p, d and f-orbitals can accommodate 6, 10 and 14 electrons respectively.
There is a trend each electron follows while filling these orbitals and it is given as-

Following this trend, we can write the electronic configuration of the +3 state of iron.
We know that the atomic number of iron is 26. In +3 state it will lose 3 electrons therefore, we will be left with 23electrons. Following the above trend, we can write the electronic configuration of $F{{e}^{3+}}$ as- $1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}3{{s}^{2}}3{{p}^{6}}3{{d}^{5}}$
We know that the atomic number of argon is 18. Therefore, electronic configuration of argon is $1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}3{{s}^{2}}3{{p}^{6}}$ .
As we know argon lies in the third period in the periodic table and iron lies in the fourth period. Therefore, we can write the electronic configuration of iron in terms of the argon as- $\left[ Ar \right]3{{d}^{5}}$ .
Therefore, the correct answer is option (A) $\left[ Ar \right]3{{d}^{5}}$.
Note: According to Pauli’s Exclusion Principle, each orbital can hold 2 electrons. s-orbital set contains one orbital, thus can hold 2 electrons. Similarly, the p-orbital set had three orbitals, thus can hold 6 electrons and d-orbital and f-orbital have five and seven orbitals thus, can hold 10 and 14 electrons respectively.
Recently Updated Pages
JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

Isoelectronic Definition in Chemistry: Meaning, Examples & Trends

Ionisation Energy and Ionisation Potential Explained

Iodoform Reactions - Important Concepts and Tips for JEE

Introduction to Dimensions: Understanding the Basics

Instantaneous Velocity Explained: Formula, Examples & Graphs

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

Derivation of Equation of Trajectory Explained for Students

Understanding the Angle of Deviation in a Prism

Other Pages
JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

CBSE Notes Class 11 Chemistry Chapter 9 - Hydrocarbons - 2025-26

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

CBSE Notes Class 11 Chemistry Chapter 8 - Organic Chemistry Some Basic Principles And Techniques - 2025-26

JEE Advanced Weightage 2025 Chapter-Wise for Physics, Maths and Chemistry




