Electromagnetic radiation with maximum wavelength is:
(A) Ultraviolet
(B) Radiowave
(C) X-ray
(D) Infrared
Answer
264.3k+ views
Hint: It is the electromagnetic radiation which is used in communication technologies such as mobile phones, Wi-Fi, television, radio and so on. Heinrich Hertz discovered this type of waves. It is used to separate electrons from atoms and molecules, chemical changes in DNA and even in cooking food at home.
Complete step by step answer:
- Clark Maxwell first told that the radio waves existed. Heinrich Hertz demonstrated this wave in the lab.
- Like other electromagnetic radiations, they also travel at the speed of the light in the vacuum.
- They are formed by the electric charges under acceleration. These electric charges flow in a special metal conductor called antenna, then transmitted through a transmitter and finally received by the radio receiver. Radio waves occurring naturally are by lightning and astronomical objects.
- Electromagnetic radiation with maximum wavelength is the radio wave.
- The frequency ranges between 300 gigahertz (GHz) to 30 hertz (Hz).
- The representation of various types of electromagnetic radiation and its relation with energy and wavelength is shown in the following figure.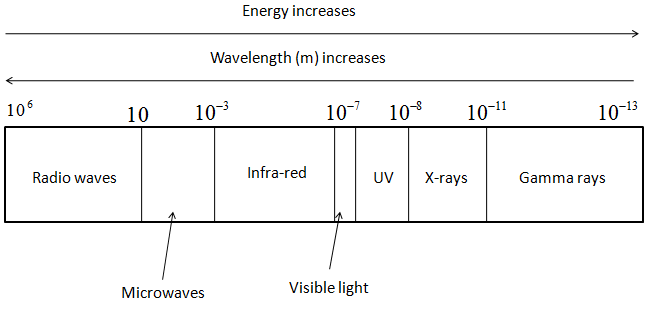
- Thus, from the figure, we can say that Radio waves will have least energy and will have the highest wavelength amongst them all.
-Nowadays, it is used as broadcasting radar, communication satellite, computer network, wireless devices such as earphones, computers and so on.
-Wi-Fi uses radio waves through wireless internet routers popularly known as LAN. It uses frequencies between 2.4 to 5.8 GHz.
So, the correct option is (B) Radiowave.
Note:
The wavelength of a wave is inversely proportional to the energy of the wave. Thus, we can say that as the wavelength of a wave increases, its energy decreases and as its wavelength decreases, its energy increases.
Complete step by step answer:
- Clark Maxwell first told that the radio waves existed. Heinrich Hertz demonstrated this wave in the lab.
- Like other electromagnetic radiations, they also travel at the speed of the light in the vacuum.
- They are formed by the electric charges under acceleration. These electric charges flow in a special metal conductor called antenna, then transmitted through a transmitter and finally received by the radio receiver. Radio waves occurring naturally are by lightning and astronomical objects.
- Electromagnetic radiation with maximum wavelength is the radio wave.
- The frequency ranges between 300 gigahertz (GHz) to 30 hertz (Hz).
- The representation of various types of electromagnetic radiation and its relation with energy and wavelength is shown in the following figure.

- Thus, from the figure, we can say that Radio waves will have least energy and will have the highest wavelength amongst them all.
-Nowadays, it is used as broadcasting radar, communication satellite, computer network, wireless devices such as earphones, computers and so on.
-Wi-Fi uses radio waves through wireless internet routers popularly known as LAN. It uses frequencies between 2.4 to 5.8 GHz.
So, the correct option is (B) Radiowave.
Note:
The wavelength of a wave is inversely proportional to the energy of the wave. Thus, we can say that as the wavelength of a wave increases, its energy decreases and as its wavelength decreases, its energy increases.
Recently Updated Pages
JEE Main Mock Test 2025-26: Principles Related To Practical

JEE Main 2025-26 Experimental Skills Mock Test – Free Practice

JEE Main 2025-26 Electronic Devices Mock Test: Free Practice Online

JEE Main 2025-26 Mock Tests: Free Practice Papers & Solutions

JEE Main 2025-26: Magnetic Effects of Current & Magnetism Mock Test

JEE Main Statistics and Probability Mock Test 2025-26

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

How to Convert a Galvanometer into an Ammeter or Voltmeter

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

JEE Advanced Marks vs Rank 2025 - Predict Your IIT Rank Based on Score

NCERT Solutions For Class 11 Chemistry In Hindi Chapter 1 Some Basic Concepts Of Chemistry - 2025-26

Understanding Electromagnetic Waves and Their Importance




