Draw the structure of a butanone molecule.
Answer
264.3k+ views
Hint: Attempt This question by breaking the given molecule to decipher its meaning. For example – methane can be broken to ‘meth-‘+ ‘-ane’, which means one carbon and alkane respectively.
Complete step by step answer:
Butanone, as the name suggests, is made up of “but- “+ “-ane” + “-one”. Therefore, we can see the following points from it –
‘but- ‘stands for four carbons.
‘-ane’ suggests that it is an alkane
‘-one’ suggests that it is a ketone.
Therefore, we can draw the structure as –
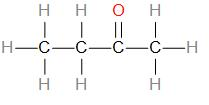
As we can see, the given compound is a ketone, therefore, double bonded oxygen can only be placed at the second carbon. The compound can also be called Methyl ethyl ketone.
We can write its compressed formula as – \[C{{H}_{3}}C{{H}_{2}}COC{{H}_{3}}\].
Butanone is an organic compound, which appears as a colorless liquid with a sharp and sweet odor. This compound is produced industrially on a large scale, but it occurs scarcely in nature. It is partially soluble in water and is commonly used as an industrial solvent. Also, it is an isomer of tetrahydrofuran.
Additional information: Root words - meth, eth, prop, but, pent are words which define the number of carbons in the compound.
Note: Butanone can be produced by oxidation of 2-butanol. The dehydrogenation of 2-butanol is catalyzed by copper, zinc, or bronze. Liquid-phase oxidation of heavy naphtha and the Fischer-Tropsch reaction, both produce mixed oxygenate streams, from which 2-butanone is extracted by fractionation.
Complete step by step answer:
Butanone, as the name suggests, is made up of “but- “+ “-ane” + “-one”. Therefore, we can see the following points from it –
‘but- ‘stands for four carbons.
‘-ane’ suggests that it is an alkane
‘-one’ suggests that it is a ketone.
Therefore, we can draw the structure as –

As we can see, the given compound is a ketone, therefore, double bonded oxygen can only be placed at the second carbon. The compound can also be called Methyl ethyl ketone.
We can write its compressed formula as – \[C{{H}_{3}}C{{H}_{2}}COC{{H}_{3}}\].
Butanone is an organic compound, which appears as a colorless liquid with a sharp and sweet odor. This compound is produced industrially on a large scale, but it occurs scarcely in nature. It is partially soluble in water and is commonly used as an industrial solvent. Also, it is an isomer of tetrahydrofuran.
Additional information: Root words - meth, eth, prop, but, pent are words which define the number of carbons in the compound.
Note: Butanone can be produced by oxidation of 2-butanol. The dehydrogenation of 2-butanol is catalyzed by copper, zinc, or bronze. Liquid-phase oxidation of heavy naphtha and the Fischer-Tropsch reaction, both produce mixed oxygenate streams, from which 2-butanone is extracted by fractionation.
Recently Updated Pages
JEE Main Mock Test 2025-26: Principles Related To Practical

JEE Main 2025-26 Experimental Skills Mock Test – Free Practice

JEE Main 2025-26 Electronic Devices Mock Test: Free Practice Online

JEE Main 2025-26 Mock Tests: Free Practice Papers & Solutions

JEE Main 2025-26: Magnetic Effects of Current & Magnetism Mock Test

JEE Main Statistics and Probability Mock Test 2025-26

Trending doubts
JEE Main Marks vs Percentile 2026: Predict Your Score Easily

JEE Main Cutoff 2026: Category-wise Qualifying Percentile

JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Marks vs Rank 2026: Expected Rank for 300 to 0 Marks

NIT Cutoff 2026: Tier-Wise Opening and Closing Ranks for B.Tech. Admission

JEE Mains 2026 Subject Wise Percentile Explained

Other Pages
Valentine Week 2026 Complete List, Dates, Quotes, Wishes & Celebration Guide

Mother's Day 2026: Date, History, Wishes, Quotes, Shayari, Gifts and Card Ideas

Navratri 2026: Day-to-Day Navratri Colours Guide with Correct Dates, Devi Names, and Significance

UP Board 10th Result 2026 OUT: Check Result, Pass Percentage, Toppers and Marksheet Download Steps

Odisha 10th Result 2026 (LIVE Soon): BSE HSC Result Link, Date & Updates

Chaitra Navratri 2026 Calendar Dates, Ghatsthapana Shuabh Muhurat Timings, Rituals, and Significance




