How does phenol react with conc. Nitric acid? Give an equation.
Answer
258.6k+ views
Hint: The reaction taking place between phenol and concentrated nitric acid is a nitration reaction. Nitration is a chemical reaction where the replacement of a Hydrogen atom present in a chemical compound is done with a nitro group.
Complete Step by Step Solution:
Phenol is an aromatic hydrocarbon where a hydroxyl group is present in a benzene ring. The molecular formula of phenol is \[{C_6}{H_5}OH\]. Phenol is a white crystalline solid which is volatile in nature.
Nitric acid is a strong acid having a molecular formula \[HN{O_3}\].
The nitration reaction is a type of electrophilic aromatic substitution where a hydrogen atom present in an aromatic hydrocarbon is displaced by an electrophile.
In the reaction between phenol and conc. Nitric acid, the nitro group is the electrophile.
In phenol, the hydroxyl group ease the delocalisation of the charge present in the aromatic compound.
When phenol reacts with concentrated nitric acid, 2, 4, 6-trinitrophenol is formed as the main product.
2, 4, 6-trinitrophenol is commonly known as Picric acid.
The reaction taking place between phenol and nitric acid is shown below.
Image: Nitration of phenol
Therefore, the product formed on the reaction of phenol and nitric acid is 2, 4, 6-trinitrophenol.
Note: When phenol is reacted with a dilute nitric acid solution at a temperature of 298K, it give a mixture of ortho and para derivative of nitrophenol as a main product. This mixture can be further separated by the distillation process due to their difference in boiling point. The ortho derivative is less volatile as compared to the para derivative due to hydrogen bonding.
The reaction is shown below.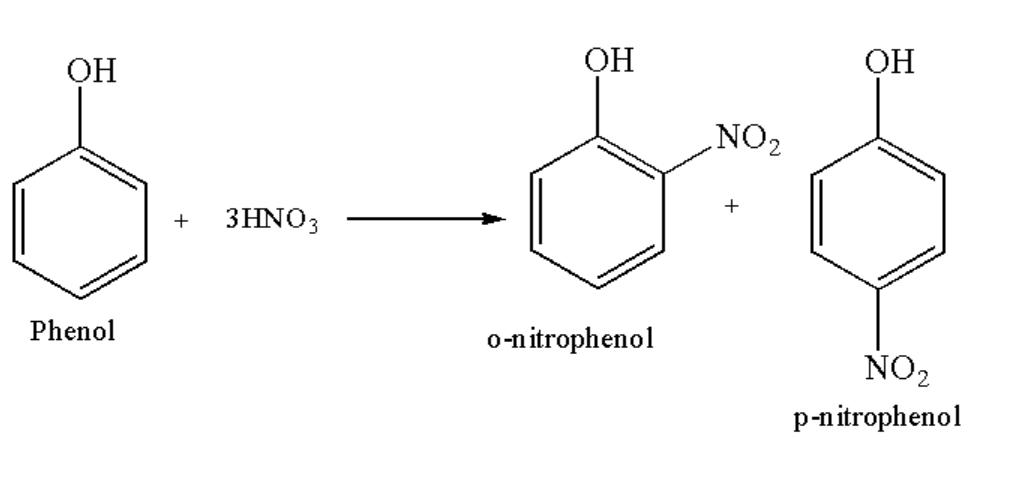
Image: Nitration of phenol
Complete Step by Step Solution:
Phenol is an aromatic hydrocarbon where a hydroxyl group is present in a benzene ring. The molecular formula of phenol is \[{C_6}{H_5}OH\]. Phenol is a white crystalline solid which is volatile in nature.
Nitric acid is a strong acid having a molecular formula \[HN{O_3}\].
The nitration reaction is a type of electrophilic aromatic substitution where a hydrogen atom present in an aromatic hydrocarbon is displaced by an electrophile.
In the reaction between phenol and conc. Nitric acid, the nitro group is the electrophile.
In phenol, the hydroxyl group ease the delocalisation of the charge present in the aromatic compound.
When phenol reacts with concentrated nitric acid, 2, 4, 6-trinitrophenol is formed as the main product.
2, 4, 6-trinitrophenol is commonly known as Picric acid.
The reaction taking place between phenol and nitric acid is shown below.
Image: Nitration of phenol
Therefore, the product formed on the reaction of phenol and nitric acid is 2, 4, 6-trinitrophenol.
Note: When phenol is reacted with a dilute nitric acid solution at a temperature of 298K, it give a mixture of ortho and para derivative of nitrophenol as a main product. This mixture can be further separated by the distillation process due to their difference in boiling point. The ortho derivative is less volatile as compared to the para derivative due to hydrogen bonding.
The reaction is shown below.

Image: Nitration of phenol
Recently Updated Pages
Disproportionation Reaction: Definition, Example & JEE Guide

Hess Law of Constant Heat Summation: Definition, Formula & Applications

JEE General Topics in Chemistry Important Concepts and Tips

JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Understanding the Different Types of Solutions in Chemistry

Other Pages
JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

JEE Advanced Weightage 2025 Chapter-Wise for Physics, Maths and Chemistry

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners




