Why distillation is a chemical reaction?
(A) Formation of products occur
(B) Change in state of reactants
(C) Both A and B
(D) Distillation is not a chemical reaction.
Answer
264.3k+ views
Hint: Distillation is a physical separation process. It is used to separate a mixture of substances by heating a liquid that undergoes a phase change; from liquid to gas. There is no involvement of reactants or products in the whole process.
Complete step-by-step answer:
Distillation refers to the selective boiling of a liquid mixture and subsequent condensation of a component in that mixture. It is a separation technique that can be used to either increase the concentration of a particular component in the mixture or to obtain pure components from the mixture. The process of distillation explains the difference in the boiling points of the components in the liquid mixture by transforming one of them into a gaseous state.
Distillation is not a chemical reaction but it can be considered as a physical separation process. Physical changes include the changes of state: solid to liquid, fusion; liquid to gas, vaporization; gas to solid, deposition. No chemical bonds are damaged in this process, other than the weak dispersion forces between molecules.
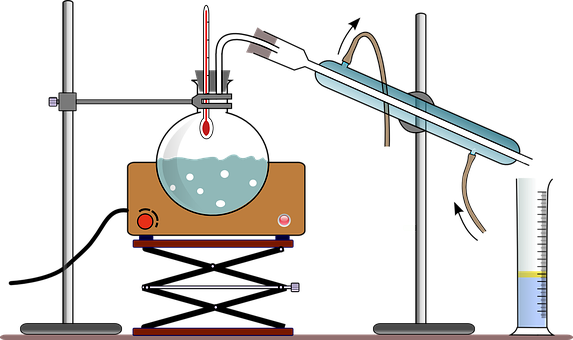
While a chemical reaction is a process in which reactants are converted into products by either bond-breaking or bond-making. As no such step occurs in the process of distillation, it is not a chemical reaction. Thus, no product is formed and no change in the state of reaction occurs.
Hence the correct option is (D).
Note: Distillation is a powerful technique used for separating the component from a miscible fluid mixture by means of selective evaporation and condensation. It is a purification method that involves boiling a liquid and purifying it by different types of distillation processes.
Complete step-by-step answer:
Distillation refers to the selective boiling of a liquid mixture and subsequent condensation of a component in that mixture. It is a separation technique that can be used to either increase the concentration of a particular component in the mixture or to obtain pure components from the mixture. The process of distillation explains the difference in the boiling points of the components in the liquid mixture by transforming one of them into a gaseous state.
Distillation is not a chemical reaction but it can be considered as a physical separation process. Physical changes include the changes of state: solid to liquid, fusion; liquid to gas, vaporization; gas to solid, deposition. No chemical bonds are damaged in this process, other than the weak dispersion forces between molecules.

While a chemical reaction is a process in which reactants are converted into products by either bond-breaking or bond-making. As no such step occurs in the process of distillation, it is not a chemical reaction. Thus, no product is formed and no change in the state of reaction occurs.
Hence the correct option is (D).
Note: Distillation is a powerful technique used for separating the component from a miscible fluid mixture by means of selective evaporation and condensation. It is a purification method that involves boiling a liquid and purifying it by different types of distillation processes.
Recently Updated Pages
JEE Main Mock Test 2025-26: Principles Related To Practical

JEE Main 2025-26 Experimental Skills Mock Test – Free Practice

JEE Main 2025-26 Electronic Devices Mock Test: Free Practice Online

JEE Main 2025-26 Mock Tests: Free Practice Papers & Solutions

JEE Main 2025-26: Magnetic Effects of Current & Magnetism Mock Test

JEE Main Statistics and Probability Mock Test 2025-26

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

How to Convert a Galvanometer into an Ammeter or Voltmeter

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

JEE Advanced Marks vs Rank 2025 - Predict Your IIT Rank Based on Score

NCERT Solutions For Class 11 Chemistry In Hindi Chapter 1 Some Basic Concepts Of Chemistry - 2025-26

Understanding Electromagnetic Waves and Their Importance




