Cyclic trimer structure of $S{{O}_{3}}$contains:
(A) Six S=O bonds and three S-O-S bonds
(B) Three S=O bonds and six S-O-S bonds
(C) Three S=O bonds and two S-O-S bonds
(D) Six S=O bonds and two S-O-S bonds
Answer
267.6k+ views
Hint: Sulphur has valency as 2 and oxygen as valency as 2. Sulphur trioxide has trigonal planar geometry. Sulphur has oxidation state +6. Structure of cyclic trimer of Sulphur trioxide is as following:
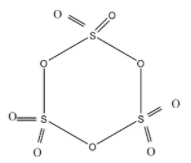
Complete step by step solution:
-Sulphur has atomic number 16. It belongs to group 16 of the periodic table. It is a p-block element. It has a valency as 2.
-Sulphur has electronic configuration as \[1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}3{{s}^{2}}3{{p}^{4}}\].
-oxygen has an atomic number 8. It belongs to group 16 of the periodic table. It has a valency of 2.
-Gaseous Sulphur trioxide has a trigonal planar structure.
-On condensation of gas, absolutely pure Sulphur trioxide undergoes condensation into a trimer.
-Sulphur atom has an oxidation state of +6.
-Sulphur trioxide is a strong Lewis acid.
-On direct oxidation of Sulphur dioxide produces Sulphur trioxide.
-Sulphur trioxide is hygroscopic.
-In sulfonation reactions, Sulphur trioxide is an essential reagent.
-Sulphur trioxide is an essential reagent in sulfonation reactions. Sulphur trioxide is generated in situ from sulphuric acid.
-The Lewis structure consists of S=O double bond and two S-O dative bonds without the utilization of d orbitals.
As shown in the diagram, the Cyclic trimer structure of $S{{O}_{3}}$contains (A) Six S=O bonds and three S-O-S bonds.
So, the correct option is (A).
Note: Pure Sulphur trioxide gas on condensation leads to the formation of the trimer. The Lewis structure consists of S=O double bond and two S-O dative bonds without the utilization of d orbitals. Gaseous Sulphur trioxide has a trigonal planar structure. On condensation of gas, absolutely pure Sulphur trioxide undergoes condensation into a trimer. Sulphur has an oxidation number of +6. The Valency of Sulphur is two.

Complete step by step solution:
-Sulphur has atomic number 16. It belongs to group 16 of the periodic table. It is a p-block element. It has a valency as 2.
-Sulphur has electronic configuration as \[1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}3{{s}^{2}}3{{p}^{4}}\].
-oxygen has an atomic number 8. It belongs to group 16 of the periodic table. It has a valency of 2.
-Gaseous Sulphur trioxide has a trigonal planar structure.
-On condensation of gas, absolutely pure Sulphur trioxide undergoes condensation into a trimer.
-Sulphur atom has an oxidation state of +6.
-Sulphur trioxide is a strong Lewis acid.
-On direct oxidation of Sulphur dioxide produces Sulphur trioxide.
-Sulphur trioxide is hygroscopic.
-In sulfonation reactions, Sulphur trioxide is an essential reagent.
-Sulphur trioxide is an essential reagent in sulfonation reactions. Sulphur trioxide is generated in situ from sulphuric acid.
-The Lewis structure consists of S=O double bond and two S-O dative bonds without the utilization of d orbitals.

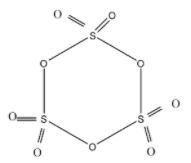
As shown in the diagram, the Cyclic trimer structure of $S{{O}_{3}}$contains (A) Six S=O bonds and three S-O-S bonds.
So, the correct option is (A).
Note: Pure Sulphur trioxide gas on condensation leads to the formation of the trimer. The Lewis structure consists of S=O double bond and two S-O dative bonds without the utilization of d orbitals. Gaseous Sulphur trioxide has a trigonal planar structure. On condensation of gas, absolutely pure Sulphur trioxide undergoes condensation into a trimer. Sulphur has an oxidation number of +6. The Valency of Sulphur is two.
Recently Updated Pages
Disproportionation Reaction: Definition, Example & JEE Guide

Hess Law of Constant Heat Summation: Definition, Formula & Applications

Algebra Made Easy: Step-by-Step Guide for Students

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

JEE Energetics Important Concepts and Tips for Exam Preparation

Chemical Properties of Hydrogen - Important Concepts for JEE Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

How to Convert a Galvanometer into an Ammeter or Voltmeter

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

JEE Advanced 2026 Marks vs Rank: Estimate IIT Rank from Your Score

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

NCERT Solutions For Class 11 Chemistry In Hindi Chapter 1 Some Basic Concepts Of Chemistry - 2025-26

Understanding Electromagnetic Waves and Their Importance




