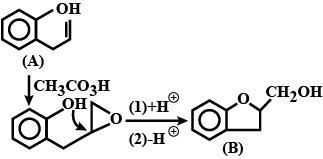
Consider the following reaction and identify (B)
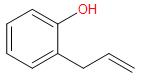 \[\underset{{{H}_{3}}O}{\overset{C{{H}_{3}}C{{O}_{3}}H}{\mathop{\to }}}\,\]\[(B)\]
\[\underset{{{H}_{3}}O}{\overset{C{{H}_{3}}C{{O}_{3}}H}{\mathop{\to }}}\,\]\[(B)\]
A. 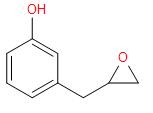
B. 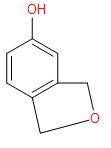
C. 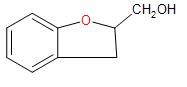
D. 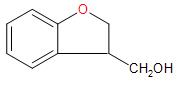
Answer
257.1k+ views
Hint: The reaction given in the question is an epoxidation reaction where an alkene is subjected to a peroxyacid to convert it into an epoxide. We can also say that epoxidation is the electrophilic addition of oxygen to the double bond of the alkene. That is what actually happens here and we must keep that in mind while solving this reaction.
Complete step by step answer:
An epoxide is a 3-membered ring containing two carbon atoms and one oxygen atom. We can consider it to be cyclic ether. It is interesting because it is easily opened due to small ring strain and due to the electronegativity of the oxygen atom.
The mechanism of this reaction is a little complex. While it is considered a single step reaction, it involves several changes. The double bond is our nucleophile and attacks the more electrophilic oxygen. This breaks the weak oxygen-oxygen bond and creates a new carbonyl. Once this carbonyl is formed, rearrangement occurs and the more electrophilic oxygen is released to become the oxygen of the epoxide.
The oxygen can only attack from one face of the alkene. This means that the stereochemistry of the alkene is retained. If we start with a cis alkene we will get a cis epoxide. If we start with a trans alkene, we will get a trans epoxide.
Hence, Option (C) is the correct answer.
Additional information:
Generally, we use peroxy acids in this electrophilic addition to the alkene. A peroxy acid is like a carboxylic acid, but has two oxygen atoms bonded to each other.
Note: This reaction is versatile, and works on many different alkenes. But we remember that the reaction will not work on the double bonds of an aromatic compound. Epoxides are essential from the examination point of view and we can expect at least one question on this.
Complete step by step answer:
An epoxide is a 3-membered ring containing two carbon atoms and one oxygen atom. We can consider it to be cyclic ether. It is interesting because it is easily opened due to small ring strain and due to the electronegativity of the oxygen atom.
The mechanism of this reaction is a little complex. While it is considered a single step reaction, it involves several changes. The double bond is our nucleophile and attacks the more electrophilic oxygen. This breaks the weak oxygen-oxygen bond and creates a new carbonyl. Once this carbonyl is formed, rearrangement occurs and the more electrophilic oxygen is released to become the oxygen of the epoxide.
The oxygen can only attack from one face of the alkene. This means that the stereochemistry of the alkene is retained. If we start with a cis alkene we will get a cis epoxide. If we start with a trans alkene, we will get a trans epoxide.

Hence, Option (C) is the correct answer.
Additional information:
Generally, we use peroxy acids in this electrophilic addition to the alkene. A peroxy acid is like a carboxylic acid, but has two oxygen atoms bonded to each other.
Note: This reaction is versatile, and works on many different alkenes. But we remember that the reaction will not work on the double bonds of an aromatic compound. Epoxides are essential from the examination point of view and we can expect at least one question on this.
Recently Updated Pages
JEE Main Mock Test 2025-26: Principles Related To Practical

JEE Main 2025-26 Mock Test: Organic Compounds Containing Nitrogen

JEE Main Mock Test 2025-26: Purification & Characterisation of Organic Compounds

JEE Main 2025-26 Mock Test: Principles Related To Practical

JEE Main Mock Test 2025-26: Principles & Best Practices

Purification and Characterisation of Organic Compounds JEE Main 2025-26 Mock Test

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

JEE Main Colleges 2026: Complete List of Participating Institutes

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Hybridisation in Chemistry – Concept, Types & Applications

Other Pages
JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 10 Biomolecules - 2025-26

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26




