Consider that a \[{d^6}\] metal ion (\[{M^{2 + }}\]) forms a complex with aqua ligands, and the spin only magnetic moment of the complex is 4.90 BM. The geometry and the crystal field stabilisation energy of the complex is:
A. Tetrahedral and \[-0.6{\Delta _t}\]
B. Tetrahedral and \[-1.6{\Delta _t} + {\rm{ }}1P\]
C. Octahedral and \[-1.6{\Delta _0}\]
D. Octahedral and \[-2.4{\Delta _0} + {\rm{ }}2P\]
Answer
257.1k+ views
Hint: From the given spin-only magnetic moment the number of unpaired electrons can be calculated. The crystal field stabilisation energy can be calculated from the d-orbital splitting pattern in tetrahedral and octahedral situations.
Complete Step by Step Solution:
The spin-only magnetic moment of a transition metal is given by the formula \[\sqrt {n(n + 2)} \] where n = the number of unpaired electrons in the valence shell of the transition metal atom. In the question, the spin-only magnetic moment is given as 4.90 BM. Therefore, we have \[\sqrt {n(n + 2)} = 4.90\]\[ \Rightarrow n = 4\]which means in the d6 metal ion there are four unpaired electrons in its valence d-orbital.
Here, the metal ion forms a complex with aqua ligands (\[{H_2}O\]) which is a comparatively strong field ligand. If the complex formed is octahedral, then the degenerate d-orbitals of the metal ion will split into two sets. Due to the repulsion of the d-orbitals lying along the coordinate axes (\[{d_{{x^2} - {y^2}}},{d_{{z^2}}}\]) with the incoming aqua ligands, energies of the \[{d_{{x^2} - {y^2}}}\]and \[{d_{{z^2}}}\]orbitals will increase comparatively. The \[{d_{{x^2} - {y^2}}}\]and \[{d_{{z^2}}}\]orbitals are called the \[{e_g}\]set while the \[{d_{xy}},{d_{yz}}{\rm{ and }}{{\rm{d}}_{xz}}\]orbitals form the \[{t_{2g}}\]set. This splitting is known as crystal field splittingand the energy separation is denoted as \[{\Delta _o}\]. The \[{e_g}\]orbitals increase in energy by \[ + 0.6{\Delta _o}\]and the \[{t_{2g}}\]orbitals decrease in energy by \[ - 0.4{\Delta _o}\].
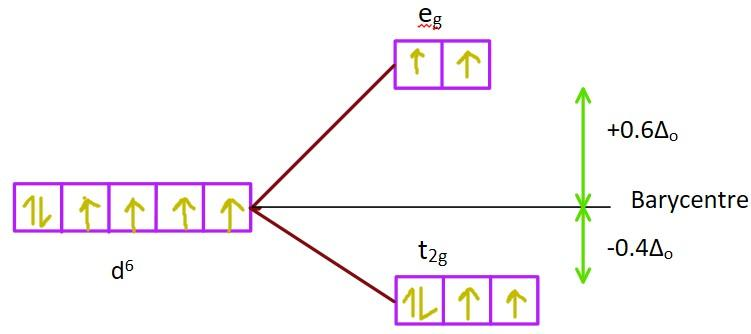
Image:d-orbital splitting in octahedral complexes
In this case, the crystal field stabilization energy (CFSE) is calculated as \[CFS{E_o} = 4 \times ( - 0.4{\Delta _o}) + 2 \times ( + 0.6{\Delta _o}) = - 0.4{\Delta _o}\]
If the complex were tetrahedral, energies of \[{d_{xy}},{d_{yz}}{\rm{ and }}{{\rm{d}}_{xz}}\]orbitals (the \[{t_2}\]set) would increase. The splitting is shown below.
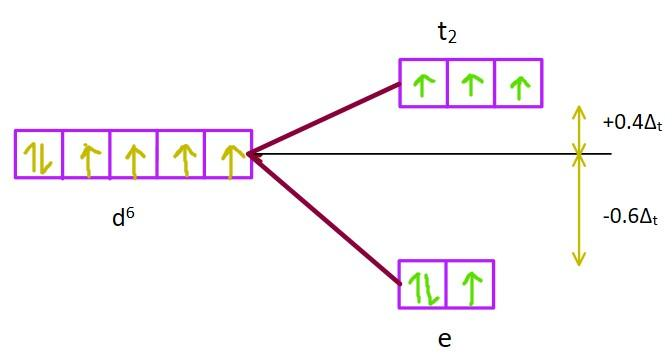
Image: d-orbital splitting in tetrahedral complexes.
In this case, the crystal field stabilization energy (CFSE) is calculated as \[CFS{E_t} = 3 \times ( - 0.6{\Delta _t}) + 3 \times ( + 0.4{\Delta _t}) = - 0.6{\Delta _t}\].
Thus, option A is correct.
Note: The order with respect to \[{\rm{[A]}}\]is \[{\rm{1}}\]and the order with respect to \[{\rm{[B]}}\] is \[{\rm{2}}\]. The overall order for the reaction will be \[(1 + 2)\, = 3\]. This implies that the reaction is of third order overall. Also, the units of rate constant (\[{\rm{k}}\]) for the given reaction can be calculated.
${\rm{rate}}\,\, = \,{\rm{k[A}}{{\rm{]}}^1}{{\rm{[B]}}^2}\\ $
$\Rightarrow {\rm{k}} = \dfrac{{{\rm{rate}}}}{{{{{\rm{[A]}}}^{\rm{1}}}{{{\rm{[B]}}}^{\rm{2}}}}}\\ \Rightarrow {\rm{k}} = \dfrac{{{\rm{mol}}{{\rm{L}}^{{\rm{ - 1}}}}{\rm{mi}}{{\rm{n}}^{{\rm{ - 1}}}}}}{{{{{\rm{(mol}}{{\rm{L}}^{{\rm{ - 1}}}}{\rm{)}}}^{\rm{1}}}{{{\rm{(mol}}{{\rm{L}}^{{\rm{ - 1}}}}{\rm{)}}}^{\rm{2}}}}}\\$
$ \Rightarrow {\rm{k}} = {\rm{mo}}{{\rm{l}}^{ - 2}}{{\rm{L}}^2}{\rm{mi}}{{\rm{n}}^{{\rm{ - 1}}}}$
The units of rate constant is found to be \[{\rm{mo}}{{\rm{l}}^{ - 2}}{{\rm{L}}^2}{\rm{mi}}{{\rm{n}}^{{\rm{ - 1}}}}\].
Complete Step by Step Solution:
The spin-only magnetic moment of a transition metal is given by the formula \[\sqrt {n(n + 2)} \] where n = the number of unpaired electrons in the valence shell of the transition metal atom. In the question, the spin-only magnetic moment is given as 4.90 BM. Therefore, we have \[\sqrt {n(n + 2)} = 4.90\]\[ \Rightarrow n = 4\]which means in the d6 metal ion there are four unpaired electrons in its valence d-orbital.
Here, the metal ion forms a complex with aqua ligands (\[{H_2}O\]) which is a comparatively strong field ligand. If the complex formed is octahedral, then the degenerate d-orbitals of the metal ion will split into two sets. Due to the repulsion of the d-orbitals lying along the coordinate axes (\[{d_{{x^2} - {y^2}}},{d_{{z^2}}}\]) with the incoming aqua ligands, energies of the \[{d_{{x^2} - {y^2}}}\]and \[{d_{{z^2}}}\]orbitals will increase comparatively. The \[{d_{{x^2} - {y^2}}}\]and \[{d_{{z^2}}}\]orbitals are called the \[{e_g}\]set while the \[{d_{xy}},{d_{yz}}{\rm{ and }}{{\rm{d}}_{xz}}\]orbitals form the \[{t_{2g}}\]set. This splitting is known as crystal field splittingand the energy separation is denoted as \[{\Delta _o}\]. The \[{e_g}\]orbitals increase in energy by \[ + 0.6{\Delta _o}\]and the \[{t_{2g}}\]orbitals decrease in energy by \[ - 0.4{\Delta _o}\].

Image:d-orbital splitting in octahedral complexes
In this case, the crystal field stabilization energy (CFSE) is calculated as \[CFS{E_o} = 4 \times ( - 0.4{\Delta _o}) + 2 \times ( + 0.6{\Delta _o}) = - 0.4{\Delta _o}\]
If the complex were tetrahedral, energies of \[{d_{xy}},{d_{yz}}{\rm{ and }}{{\rm{d}}_{xz}}\]orbitals (the \[{t_2}\]set) would increase. The splitting is shown below.

Image: d-orbital splitting in tetrahedral complexes.
In this case, the crystal field stabilization energy (CFSE) is calculated as \[CFS{E_t} = 3 \times ( - 0.6{\Delta _t}) + 3 \times ( + 0.4{\Delta _t}) = - 0.6{\Delta _t}\].
Thus, option A is correct.
Note: The order with respect to \[{\rm{[A]}}\]is \[{\rm{1}}\]and the order with respect to \[{\rm{[B]}}\] is \[{\rm{2}}\]. The overall order for the reaction will be \[(1 + 2)\, = 3\]. This implies that the reaction is of third order overall. Also, the units of rate constant (\[{\rm{k}}\]) for the given reaction can be calculated.
${\rm{rate}}\,\, = \,{\rm{k[A}}{{\rm{]}}^1}{{\rm{[B]}}^2}\\ $
$\Rightarrow {\rm{k}} = \dfrac{{{\rm{rate}}}}{{{{{\rm{[A]}}}^{\rm{1}}}{{{\rm{[B]}}}^{\rm{2}}}}}\\ \Rightarrow {\rm{k}} = \dfrac{{{\rm{mol}}{{\rm{L}}^{{\rm{ - 1}}}}{\rm{mi}}{{\rm{n}}^{{\rm{ - 1}}}}}}{{{{{\rm{(mol}}{{\rm{L}}^{{\rm{ - 1}}}}{\rm{)}}}^{\rm{1}}}{{{\rm{(mol}}{{\rm{L}}^{{\rm{ - 1}}}}{\rm{)}}}^{\rm{2}}}}}\\$
$ \Rightarrow {\rm{k}} = {\rm{mo}}{{\rm{l}}^{ - 2}}{{\rm{L}}^2}{\rm{mi}}{{\rm{n}}^{{\rm{ - 1}}}}$
The units of rate constant is found to be \[{\rm{mo}}{{\rm{l}}^{ - 2}}{{\rm{L}}^2}{\rm{mi}}{{\rm{n}}^{{\rm{ - 1}}}}\].
Recently Updated Pages
JEE General Topics in Chemistry Important Concepts and Tips

JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Electricity and Magnetism Explained: Key Concepts & Applications

JEE Energetics Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

JEE Main Colleges 2026: Complete List of Participating Institutes

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Hybridisation in Chemistry – Concept, Types & Applications

Other Pages
JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 10 Biomolecules - 2025-26

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26




