Chlorobenzene can be prepared by reacting aniline with:
(A) Hydrochloric acid (HCl)
(B) Cuprous chloride $\left( C{{u}_{2}}C{{l}_{2}} \right)$
(C) Chlorine in the presence of anhydrous aluminium chloride
(D) Nitrous acid followed by heating with cuprous chloride (CuCl)
Answer
264.3k+ views
Hint: Start by drawing the structure of the reactant. Think what kind of reactions aniline undergoes with the given reagents in the options. The product formed should not contain any other substituent on the ring except chlorine.
Complete step by step solution:
-Aniline is an aromatic amine. It is a base.
-Let’s have a look at the question. We need to prepare chlorobenzene from aniline. That means the amine group needs to be replaced by a chlorine group.
-We have studies that amine group can only be replaced after it has been converted to diazonium salt form, which is unstable. Direct halogenation of aniline will give rise to halogen derivatives of aniline.
-If HCl is added to aniline, then aniline being a base will abstract the proton and form anilinium chloride salt.
-When aniline is treated with nitrous acid, which is produced in situ by sodium nitrite and hydrochloric acid, at low temperature $\left( 0{}^\circ C-5{}^\circ C \right)$, then aniline is converted to a benzene diazonium salt. This reaction is known as diazotization.
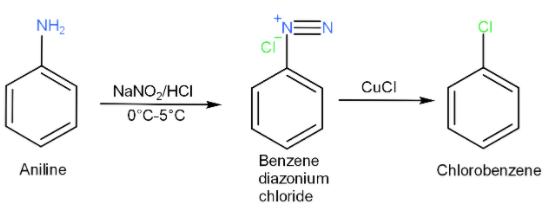
-When a freshly prepared solution of benzene diazonium salt is treated with cuprous chloride (CuCl), it gives chlorobenzene. This reaction is known as Sandmeyer’s reaction.
-Therefore, chlorobenzene can be prepared by reacting aniline with nitrous acid followed by heating with cuprous chloride (CuCl).
Therefore, the correct option is (D).
Note: Chlorobenzene can also be prepared by treating benzene diazonium salt with copper powder and hydrochloric acid. This reaction is known as Gattermann reaction and has a better yield than Sandmeyer reaction. Remember in aromatic compounds electrophilic substitution reactions occur readily and therefore, direct halogenation will yield halo-substituted compounds.
Complete step by step solution:
-Aniline is an aromatic amine. It is a base.
-Let’s have a look at the question. We need to prepare chlorobenzene from aniline. That means the amine group needs to be replaced by a chlorine group.
-We have studies that amine group can only be replaced after it has been converted to diazonium salt form, which is unstable. Direct halogenation of aniline will give rise to halogen derivatives of aniline.
-If HCl is added to aniline, then aniline being a base will abstract the proton and form anilinium chloride salt.
-When aniline is treated with nitrous acid, which is produced in situ by sodium nitrite and hydrochloric acid, at low temperature $\left( 0{}^\circ C-5{}^\circ C \right)$, then aniline is converted to a benzene diazonium salt. This reaction is known as diazotization.

-When a freshly prepared solution of benzene diazonium salt is treated with cuprous chloride (CuCl), it gives chlorobenzene. This reaction is known as Sandmeyer’s reaction.
-Therefore, chlorobenzene can be prepared by reacting aniline with nitrous acid followed by heating with cuprous chloride (CuCl).
Therefore, the correct option is (D).
Note: Chlorobenzene can also be prepared by treating benzene diazonium salt with copper powder and hydrochloric acid. This reaction is known as Gattermann reaction and has a better yield than Sandmeyer reaction. Remember in aromatic compounds electrophilic substitution reactions occur readily and therefore, direct halogenation will yield halo-substituted compounds.
Recently Updated Pages
JEE Main Mock Test 2025-26: Principles Related To Practical

JEE Main 2025-26 Organic Compounds Containing Nitrogen Mock Test

JEE Main Chemical Kinetics Mock Test 2025-26: Free Practice Online

JEE Main 2025-26 Organic Compounds Containing Oxygen Mock Test

JEE Main 2025-26 Mock Test: Organic Compounds Containing Oxygen

JEE Main 2025-26 Organic Compounds Containing Halogens Mock Test

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

How to Convert a Galvanometer into an Ammeter or Voltmeter

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




