Chiral carbon is:
(A) carbon in which all four valencies are satisfied with four different atoms or group of atoms
(B) carbon in which at least two of their valency are satisfied with four different atoms or group of atoms
(C) carbon in which all four valencies satisfied with same atoms
(D) none of these
Answer
256.2k+ views
Hint: A chiral carbon indicates the presence of optical activity around the central carbon. The mirror image of an optically active compound should not superimpose over the original image. So, if the same groups or atoms are attached the mirror image will not be unique.
Complete step-by-step answer:
The word “isomer” is derived from the Greek words "isos" and "mers". "Isos" means equal and "mers" means parts, so "isomers" means equal parts.
Isomerism is the phenomenon in which two or more compounds have the same chemical formula but differ in chemical structures. Chemical compounds that have identical chemical formulas but differ in properties and the arrangement of atoms in the molecule are called isomers i.e. they exhibit isomerism.
Isomerism is of two types namely, Structural isomerism and stereoisomerism.
In stereoisomerism, the compounds have the same chemical formula but differ in their respective orientations of the atoms belonging to the compound in a 3D space.
The types of stereoisomerism are:
- Geometrical
- Optical
Optical isomers are two compounds having the same molecular formula but differ in their spatial arrangements of atoms, which have non-superimposable mirror images.
Since the mirror images are not the same, the groups around the chiral carbon must be different in order to satisfy the above condition. So Chiral carbon is carbon in which all four valencies are satisfied with four different atoms or groups of atoms.
Therefore, the correct answer is option (A).
Note: Geometrical isomerism is popularly known as cis-trans isomerism.
Geometrical isomers have different spatial arrangements of atoms present in the compound in a 3D space. Given below is an example of pair of isomers exhibiting geometrical isomerism:
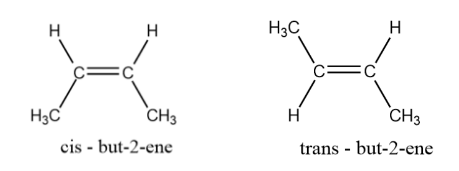
Complete step-by-step answer:
The word “isomer” is derived from the Greek words "isos" and "mers". "Isos" means equal and "mers" means parts, so "isomers" means equal parts.
Isomerism is the phenomenon in which two or more compounds have the same chemical formula but differ in chemical structures. Chemical compounds that have identical chemical formulas but differ in properties and the arrangement of atoms in the molecule are called isomers i.e. they exhibit isomerism.
Isomerism is of two types namely, Structural isomerism and stereoisomerism.
In stereoisomerism, the compounds have the same chemical formula but differ in their respective orientations of the atoms belonging to the compound in a 3D space.
The types of stereoisomerism are:
- Geometrical
- Optical
Optical isomers are two compounds having the same molecular formula but differ in their spatial arrangements of atoms, which have non-superimposable mirror images.
Since the mirror images are not the same, the groups around the chiral carbon must be different in order to satisfy the above condition. So Chiral carbon is carbon in which all four valencies are satisfied with four different atoms or groups of atoms.
Therefore, the correct answer is option (A).
Note: Geometrical isomerism is popularly known as cis-trans isomerism.
Geometrical isomers have different spatial arrangements of atoms present in the compound in a 3D space. Given below is an example of pair of isomers exhibiting geometrical isomerism:

Recently Updated Pages
Electricity and Magnetism Explained: Key Concepts & Applications

JEE Energetics Important Concepts and Tips for Exam Preparation

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

JEE Main 2023 (February 1st Shift 1) Maths Question Paper with Answer Key

JEE Main 2023 (February 1st Shift 2) Maths Question Paper with Answer Key

JEE Main 2023 (February 1st Shift 2) Chemistry Question Paper with Answer Key

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

JEE Main Colleges 2026: Complete List of Participating Institutes

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Hybridisation in Chemistry – Concept, Types & Applications

Other Pages
JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

CBSE Notes Class 11 Chemistry Chapter 9 - Hydrocarbons - 2025-26

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

JEE Advanced Weightage 2025 Chapter-Wise for Physics, Maths and Chemistry

CBSE Notes Class 11 Chemistry Chapter 8 - Organic Chemistry Some Basic Principles And Techniques - 2025-26




