Benzaldehyde condenses with N, N-dimethylaniline in presence of anhydrous \[ZnC{l_2}\] to give
A. Michler’s ketone
B. Azo dye
C. Malachite green
D. Buffer yellow
Answer
267k+ views
Hint: The chemical formula of benzaldehyde is \[{C_6}{H_5}CH = O\]. In N, N-dimethylaniline and hydrogen of the amine group are replaced by two methyl groups. In the given reaction dehydration takes place as a water molecule is removed. It is a type of condensation reaction.
Complete Step by Step Solution:
Benzaldehyde in reaction with N,N-dimethyl aniline in presence of anhydrous zinc chloride gives malachite green. Anhydrous means free from water. Here, the two hydrogen atoms from two molecules of N, N-dimethylaniline are removed along with one oxygen atom of benzaldehyde forming a malachite green and removing the water molecule.
The complete reaction between benzaldehyde and N, N-dimethylaniline in presence of anhydrous \[ZnC{l_2}\]is shown below.
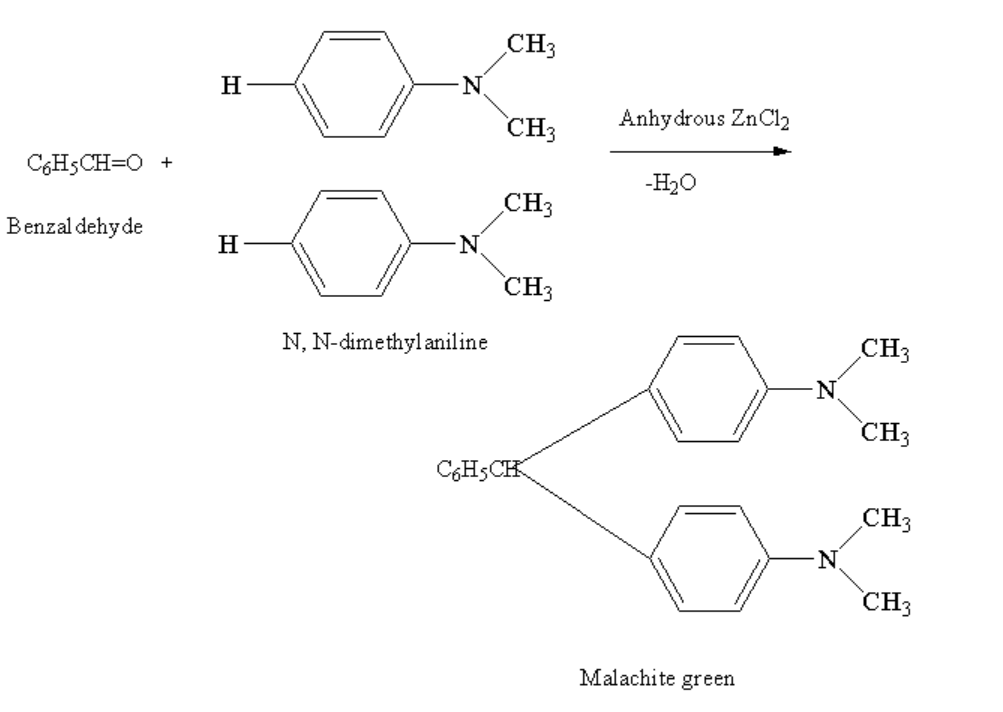
Image: Reaction of benzaldehyde and N, N-Dimethylaniline
Therefore, the correct option is C.
Additional information: Hermann Fisher was the first scientist to prepare Leuco malachite green in the year 1877. Malachite green is generally used in the dyestuff industry as triarylmethane and in the pigment industry. The term malachite green is used due to its coloured green cation. Malachite green is very hard to detect as it does not give any fluorescence in an aqueous solution.
Note: It should be noted that two molecules of N, N-dimethylaniline are reacted not one molecule with benzaldehyde to give malachite green. First when benzaldehyde reacts with N, N-dimethylaniline it gives leuco malachite green which is a colourless compound which further on reacting gives hydrogen chloride and oxidation forms malachite green.
Complete Step by Step Solution:
Benzaldehyde in reaction with N,N-dimethyl aniline in presence of anhydrous zinc chloride gives malachite green. Anhydrous means free from water. Here, the two hydrogen atoms from two molecules of N, N-dimethylaniline are removed along with one oxygen atom of benzaldehyde forming a malachite green and removing the water molecule.
The complete reaction between benzaldehyde and N, N-dimethylaniline in presence of anhydrous \[ZnC{l_2}\]is shown below.

Image: Reaction of benzaldehyde and N, N-Dimethylaniline
Therefore, the correct option is C.
Additional information: Hermann Fisher was the first scientist to prepare Leuco malachite green in the year 1877. Malachite green is generally used in the dyestuff industry as triarylmethane and in the pigment industry. The term malachite green is used due to its coloured green cation. Malachite green is very hard to detect as it does not give any fluorescence in an aqueous solution.
Note: It should be noted that two molecules of N, N-dimethylaniline are reacted not one molecule with benzaldehyde to give malachite green. First when benzaldehyde reacts with N, N-dimethylaniline it gives leuco malachite green which is a colourless compound which further on reacting gives hydrogen chloride and oxidation forms malachite green.
Recently Updated Pages
States of Matter Chapter For JEE Main Chemistry

Classification of Drugs in Chemistry: Types, Examples & Exam Guide

Types of Solutions in Chemistry: Explained Simply

Difference Between Alcohol and Phenol: Structure, Tests & Uses

[Awaiting the three content sources: Ask AI Response, Competitor 1 Content, and Competitor 2 Content. Please provide those to continue with the analysis and optimization.]

Sign up for JEE Main 2026 Live Classes - Vedantu

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




