Assertion:In electrolytic refining of metal, impure metal is made cathode while a strip of pure metal is used as anode.
Reason: The pure metal gets deposited at anode as anode mud.
(A) Both assertion and reason are correct and reason is the correct explanation of assertion.
(B) Both assertion and Reason are correct but reason is not the correct explanation for assertion.
(C) Assertion is correct but reason is incorrect.
(D) Both assertion and reason are incorrect.
Answer
249.3k+ views
Hint: Electrolytic refining process is used for purification of metal. Metals obtained after smelting or other methods may contain impurities like unreacted oxides, other metals, non- metals and gases.
The purification of crude metals by removing metallic and non- metallic impurities is known as refining.
Complete step by step answer:
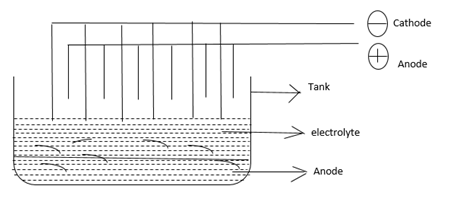
In electrolytic refining, anode [positively charged] and cathode [negatively charged] electrode is dipped into aqueous solution of their salt.
The rod of impure metal is used as anode and thin sheet of pure metal as cathode.
During electrolysis, metal from anode dissolve in the solution while the same amount of pure metal deposits on cathode.
The impurities like more reactive metals dissolve in the solution.
Less reactive metals are insoluble. They form anode mud at the bottom.
In this process $99.99\% $ pure metal is obtained.
Metals like $Ni,Al,Zn$ are refined by his method.
In the given question:
Assertion is – impure metal is made cathode while a strip of pure metal is used as anode.
But as we already discussed, impure metal is made anode and pure metal is used as cathode.
Reason is – The pure metal gets deposited at anode mud.
But pure metal gets deposited at cathode and impurities form anode mud.
Therefore, the above explanation, the correct option is [D] Both assertion and reason are incorrect.
Note: Impure metal is made anode and pure metal strip is made cathode during electrolytic refining. During electrolysis pure metal from impure anode gets dissolved in the solution. As metal is always positively charged, it is migrated towards cathode and deposit. then cathodes become thick and replaced from time to time.
The purification of crude metals by removing metallic and non- metallic impurities is known as refining.
Complete step by step answer:
In electrolytic refining, anode [positively charged] and cathode [negatively charged] electrode is dipped into aqueous solution of their salt.
The rod of impure metal is used as anode and thin sheet of pure metal as cathode.
During electrolysis, metal from anode dissolve in the solution while the same amount of pure metal deposits on cathode.
The impurities like more reactive metals dissolve in the solution.
Less reactive metals are insoluble. They form anode mud at the bottom.
In this process $99.99\% $ pure metal is obtained.
Metals like $Ni,Al,Zn$ are refined by his method.

In the given question:
Assertion is – impure metal is made cathode while a strip of pure metal is used as anode.
But as we already discussed, impure metal is made anode and pure metal is used as cathode.
Reason is – The pure metal gets deposited at anode mud.
But pure metal gets deposited at cathode and impurities form anode mud.
Therefore, the above explanation, the correct option is [D] Both assertion and reason are incorrect.
Note: Impure metal is made anode and pure metal strip is made cathode during electrolytic refining. During electrolysis pure metal from impure anode gets dissolved in the solution. As metal is always positively charged, it is migrated towards cathode and deposit. then cathodes become thick and replaced from time to time.
Recently Updated Pages
JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

Isoelectronic Definition in Chemistry: Meaning, Examples & Trends

Ionisation Energy and Ionisation Potential Explained

Iodoform Reactions - Important Concepts and Tips for JEE

Introduction to Dimensions: Understanding the Basics

Instantaneous Velocity Explained: Formula, Examples & Graphs

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

Hybridisation in Chemistry – Concept, Types & Applications

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Other Pages
CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 10 Biomolecules - 2025-26

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




