Assertion: Zinc blende and wurtzite both have fcc arrangement of \[{{\text{S}}^{2 - }}\] ions.
Reason: A unit cell of both has four formula units ${\text{ZnS}}$
(A) Both assertion and reason are correct and reason is the correct explanation of assertion
(B) Both assertion and reason are correct but reason is not the correct explanation of assertion.
(C) Assertion is correct but reason is not correct.
(D) Assertion is not correct but reason is correct.
(E) Both assertion and reason are not correct.
Answer
249.9k+ views
Hint: Zinc blend has the formula ${\text{ZnS}}$. In a zinc blend structure, ${\text{Z}}{{\text{n}}^{{\text{2 + }}}}$ and ${{\text{S}}^{2 - }}$ ions are arranged in a crystal lattice. Wurtzite has the formula ${\text{(Zn,Fe)S}}$.
Complete step by step answer:
A closely packed cubic arrangement in which the facial positions are equivalent to each of the eight corners is known as a face centred cubic arrangement.
A closely packed arrangement in two dimensions in which each sphere has six nearest neighbours and has a hexagonal symmetry is known as a hexagonal close packing arrangement.
Zinc blend has a face centred cubic (fcc) arrangement of ${{\text{S}}^{2 - }}$ ions. Wurtzite has hexagonal close packing (hcp) arrangement of ${{\text{S}}^{2 - }}$ ions.
The smallest repeating unit of the structure in a crystal lattice is known as unit cell.
The unit cell of zinc blend has four formula units of ${\text{ZnS}}$. The unit cell of wurtzite has six formula units of ${\text{ZnS}}$.
The structures of zinc blende and wurtzite are as follows:
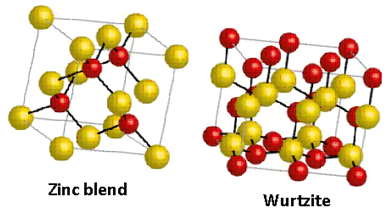
Thus, the assertion and reason both are not correct.
Thus, the correct option is (E) both assertion and reason are not correct.
Additional Information:
There are three types of unit cells:
1. Primitive cubic unit cell: The atoms are present at the eight corners of a cube.
2. Body centred cubic unit cell: The atoms are present at the eight corners of a cube and one atom is present at the centre of the structure.
3. Face centred cubic unit cell: The atoms are present at the eight corners of a cube and at the centre of all the six faces of a cube.
Note: In hcp, there are two layers of ions or atoms while in fcc there are three layers. The hcp structure has two planes while the fcc structure has three planes. The coordination number of hcp structure is $12$ and that of fcc structure is also $12$.
Complete step by step answer:
A closely packed cubic arrangement in which the facial positions are equivalent to each of the eight corners is known as a face centred cubic arrangement.
A closely packed arrangement in two dimensions in which each sphere has six nearest neighbours and has a hexagonal symmetry is known as a hexagonal close packing arrangement.
Zinc blend has a face centred cubic (fcc) arrangement of ${{\text{S}}^{2 - }}$ ions. Wurtzite has hexagonal close packing (hcp) arrangement of ${{\text{S}}^{2 - }}$ ions.
The smallest repeating unit of the structure in a crystal lattice is known as unit cell.
The unit cell of zinc blend has four formula units of ${\text{ZnS}}$. The unit cell of wurtzite has six formula units of ${\text{ZnS}}$.
The structures of zinc blende and wurtzite are as follows:

Thus, the assertion and reason both are not correct.
Thus, the correct option is (E) both assertion and reason are not correct.
Additional Information:
There are three types of unit cells:
1. Primitive cubic unit cell: The atoms are present at the eight corners of a cube.
2. Body centred cubic unit cell: The atoms are present at the eight corners of a cube and one atom is present at the centre of the structure.
3. Face centred cubic unit cell: The atoms are present at the eight corners of a cube and at the centre of all the six faces of a cube.
Note: In hcp, there are two layers of ions or atoms while in fcc there are three layers. The hcp structure has two planes while the fcc structure has three planes. The coordination number of hcp structure is $12$ and that of fcc structure is also $12$.
Recently Updated Pages
JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

Isoelectronic Definition in Chemistry: Meaning, Examples & Trends

Ionisation Energy and Ionisation Potential Explained

Iodoform Reactions - Important Concepts and Tips for JEE

Introduction to Dimensions: Understanding the Basics

Instantaneous Velocity Explained: Formula, Examples & Graphs

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

Hybridisation in Chemistry – Concept, Types & Applications

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Other Pages
CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 10 Biomolecules - 2025-26

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




