Aspirin is known as:
(A) Acetylsalicylic acid
(B) Phenyl salicylate
(C) Acetyl salicylate
(D) Methylsalicylic acid
Answer
267.9k+ views
Hint: The structure of aspirin consists of benzene ring ${ C }_{ 6 }{ H }_{ 6 }$ with carboxylic acid (COOH) and an ester as a functional group. It is prepared by Kolbe Schmitt reaction by treating phenol with an alkali. After that, an anhydride and sulphuric acid is used for the formation of aspirin.
Complete step-by-step answer:
The chemical name of aspirin is ${ 2 }$-acetoxy benzoic acid. Aspirin, otherwise called acetylsalicylic corrosive (ASA), is a drug used to treat torment, fever, and irritation. Explicit provocative conditions in which it is utilized incorporate Kawasaki malady, pericarditis, and rheumatic fever.
Anti-inflammatory medicine given soon after a coronary episode diminishes the danger of death. Ibuprofen is additionally utilized long haul to help forestall coronary episodes, strokes, and blood clumps, in individuals at high hazard. Headache medicine may likewise diminish the danger of specific sorts of disease, especially colorectal malignant growth. For agony or fever, impacts regularly start inside ${ 30 }$ minutes.
Aspirin is a nonsteroidal anti-inflammatory drug (NSAID) and works similarly to other NSAIDs but it is also antiplatelet and suppresses the normal functioning of platelets.
Acetylsalicylic acid (aspirin) is prepared from phenol by the first carboxylation of sodium phenoxide by Kolbe-Schmitt reaction to form salicylic acid followed by acetylation.
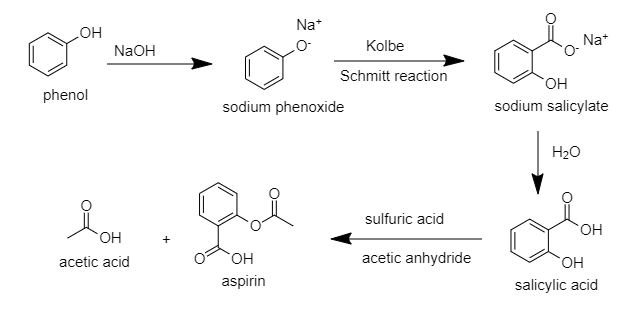
Hence, the correct option is A.
Additional Information:
When our body is in stress it triggers a reaction to the cellular level. This reaction often ends with the generation of a class of compounds called prostaglandins. It causes pain and fever in the body. Aspirin prevents the generation of prostaglandins by inhibiting the enzyme which produces them (cyclooxygenase), thus relieving pain, fever, and inflammation.
Note: The possibility to make a mistake is that you may choose option C. But aspirin is acetylsalicylic acid, not acetylsalicylate, and it is an analgesic.
Complete step-by-step answer:
The chemical name of aspirin is ${ 2 }$-acetoxy benzoic acid. Aspirin, otherwise called acetylsalicylic corrosive (ASA), is a drug used to treat torment, fever, and irritation. Explicit provocative conditions in which it is utilized incorporate Kawasaki malady, pericarditis, and rheumatic fever.
Anti-inflammatory medicine given soon after a coronary episode diminishes the danger of death. Ibuprofen is additionally utilized long haul to help forestall coronary episodes, strokes, and blood clumps, in individuals at high hazard. Headache medicine may likewise diminish the danger of specific sorts of disease, especially colorectal malignant growth. For agony or fever, impacts regularly start inside ${ 30 }$ minutes.
Aspirin is a nonsteroidal anti-inflammatory drug (NSAID) and works similarly to other NSAIDs but it is also antiplatelet and suppresses the normal functioning of platelets.
Acetylsalicylic acid (aspirin) is prepared from phenol by the first carboxylation of sodium phenoxide by Kolbe-Schmitt reaction to form salicylic acid followed by acetylation.

Hence, the correct option is A.
Additional Information:
When our body is in stress it triggers a reaction to the cellular level. This reaction often ends with the generation of a class of compounds called prostaglandins. It causes pain and fever in the body. Aspirin prevents the generation of prostaglandins by inhibiting the enzyme which produces them (cyclooxygenase), thus relieving pain, fever, and inflammation.
Note: The possibility to make a mistake is that you may choose option C. But aspirin is acetylsalicylic acid, not acetylsalicylate, and it is an analgesic.
Recently Updated Pages
Algebra Made Easy: Step-by-Step Guide for Students

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

JEE Energetics Important Concepts and Tips for Exam Preparation

Chemical Properties of Hydrogen - Important Concepts for JEE Exam Preparation

JEE General Topics in Chemistry Important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

How to Convert a Galvanometer into an Ammeter or Voltmeter

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




