An acidified solution of potassium chromate was layered with an equal volume of amyl alcohol. When it was shaken after the addition of 1 mL of 3% \[{{\mathop{\rm H}\nolimits} _2}{O_2}\], a blue alcohol layer was obtained. The blue colour is due to the formation of a chromium (VI) compound 'X'. What is the number of oxygen atoms bonded to chromium through only single bonds in a molecule of X?
Answer
259.2k+ views
Hint: Potassium dichromate is a powerful oxidising agent. It reacts with Hydrogen peroxide to give a deep blue colouration due to the formation of the Peroxo compound.
Complete Step by Step Solution:
Chromium is a transition metal of the 3d-series.
Its atomic number is 25.
Its electronic configuration is \[\left[ {{\rm{Ar}}} \right]{\rm{3}}{{\rm{d}}^{\rm{5}}}{\rm{4}}{{\rm{s}}^{\rm{2}}}\].
It shows a variable oxidation state.
The most common oxidation states are +3 and +6.
Potassium dichromate is an important compound of Chromium used as an oxidising agent.
Potassium dichromate is in the form of orange crystals.
The oxidation state of Chromium in this compound is +6.
When an acidified solution of Potassium reacts with Hydrogen peroxide, it gives a blue colouration.
This is due to the formation of peroxo compound, \[{\rm{Cr}}{\left( {{{\rm{O}}_{\rm{2}}}} \right)_{\rm{2}}}\].
.\[{{\mathop{\rm Cr}\nolimits} _2}{O_7}^{2 - } + 2{H^ + } + 4{H_2}{O_2} \to 2Cr{O_5} + 5{H_2}O\].
The structure of the product formed is
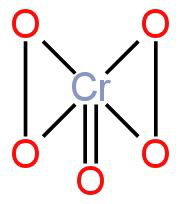
Image: Chromium pentoxide
The oxidation state of Chromium in this compound is +6.
It has a trigonal bipyramidal structure.
The number of oxygen atoms bonded to chromium only through single bonds in Chromium pentoxide is 4.
So,the number of oxygen atoms bonded to chromium only through single bonds in Chromium pentoxide is 4.
Additional Information: Chromium pentoxide decomposes to Cr3+ ions and oxygen which leads to the discoloration of the blue colour solution. It is a very good oxidising agent due to the presence of two reactive peroxo ligands. Ketones and primary alcohols when treated with it get oxidised to aldehydes and carboxylic acids.
Note: Chromium pentoxide is formed as a product when Potassium dichromate reacts with Hydrogen peroxide. This compound is a peroxo compound having one double bond and four single bonds attached to oxygen atoms. The structure of this compound is also called butterfly structure.
Complete Step by Step Solution:
Chromium is a transition metal of the 3d-series.
Its atomic number is 25.
Its electronic configuration is \[\left[ {{\rm{Ar}}} \right]{\rm{3}}{{\rm{d}}^{\rm{5}}}{\rm{4}}{{\rm{s}}^{\rm{2}}}\].
It shows a variable oxidation state.
The most common oxidation states are +3 and +6.
Potassium dichromate is an important compound of Chromium used as an oxidising agent.
Potassium dichromate is in the form of orange crystals.
The oxidation state of Chromium in this compound is +6.
When an acidified solution of Potassium reacts with Hydrogen peroxide, it gives a blue colouration.
This is due to the formation of peroxo compound, \[{\rm{Cr}}{\left( {{{\rm{O}}_{\rm{2}}}} \right)_{\rm{2}}}\].
.\[{{\mathop{\rm Cr}\nolimits} _2}{O_7}^{2 - } + 2{H^ + } + 4{H_2}{O_2} \to 2Cr{O_5} + 5{H_2}O\].
The structure of the product formed is

Image: Chromium pentoxide
The oxidation state of Chromium in this compound is +6.
It has a trigonal bipyramidal structure.
The number of oxygen atoms bonded to chromium only through single bonds in Chromium pentoxide is 4.
So,the number of oxygen atoms bonded to chromium only through single bonds in Chromium pentoxide is 4.
Additional Information: Chromium pentoxide decomposes to Cr3+ ions and oxygen which leads to the discoloration of the blue colour solution. It is a very good oxidising agent due to the presence of two reactive peroxo ligands. Ketones and primary alcohols when treated with it get oxidised to aldehydes and carboxylic acids.
Note: Chromium pentoxide is formed as a product when Potassium dichromate reacts with Hydrogen peroxide. This compound is a peroxo compound having one double bond and four single bonds attached to oxygen atoms. The structure of this compound is also called butterfly structure.
Recently Updated Pages
Disproportionation Reaction: Definition, Example & JEE Guide

Hess Law of Constant Heat Summation: Definition, Formula & Applications

JEE General Topics in Chemistry Important Concepts and Tips

JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Understanding the Different Types of Solutions in Chemistry

Other Pages
JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

JEE Advanced Weightage 2025 Chapter-Wise for Physics, Maths and Chemistry

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners




