A detergent is:
A. Cleaning agent
B. Drugs
C. Soap
D. Catalyst
Answer
249k+ views
Hint: A detergent or synthetic detergent or a syndet is a soapless soap which means it has all the properties of soap, but does not contain any soap.
They are a class of surfactants which are chemical substances that concentrate at the surface of the solution, form surface films, reduce surface tension of the solution and emulsify grease.
Complete step by step answer:
Detergents are actually ammonium, sulfate or sulphate salts of long chain hydrocarbons which contain 12 to 18 carbon atoms. But the more common detergents are the sodium salts of long chain sulphonic acids.
Detergents are surface active agents and they can remove dirt and dust by emulsifying grease. So, they act as cleansing agents. The molecule of a detergent consists of two characteristic groups, one of which is water soluble or hydrophilic and the other is oil soluble or lipophilic.
For example, sodium lauryl sulphate is a detergent. It consists of sodium ion and lauryl sulphate anion.
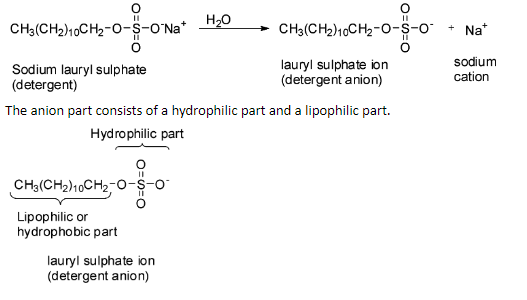
When a detergent is applied on a cloth with a greasy stain, the detergent reduces the surface tension of the water due to which the surface of the cloth is wetted thoroughly. The lipophilic parts of the detergent anion will be soluble in grease and the hydrophilic parts will be soluble in water.
When the cloth is scrubbed, the grease is pulled away from the cloth and broken into smaller droplets. Repulsion between the droplets will cause them to be suspended in water, thus forming an emulsion. Thus, the droplets will not coagulate or redeposit on the cloth. As a result, rinsing will wash away the droplets making the cloth clean.
Hence, the correct option is A.
Note:
An advantage of detergent over soap is that detergents can be used even in hard water without any wastage but some of the soap gets wasted in hard water.
However, a disadvantage of detergents is that detergents having highly branched hydrocarbon chains cause pollution in rivers. This is because the side chains stop bacteria from attacking and breaking the chains which results in slow degradation of the detergent molecule leading to their accumulation in rivers. This causes water pollution.
They are a class of surfactants which are chemical substances that concentrate at the surface of the solution, form surface films, reduce surface tension of the solution and emulsify grease.
Complete step by step answer:
Detergents are actually ammonium, sulfate or sulphate salts of long chain hydrocarbons which contain 12 to 18 carbon atoms. But the more common detergents are the sodium salts of long chain sulphonic acids.
Detergents are surface active agents and they can remove dirt and dust by emulsifying grease. So, they act as cleansing agents. The molecule of a detergent consists of two characteristic groups, one of which is water soluble or hydrophilic and the other is oil soluble or lipophilic.
For example, sodium lauryl sulphate is a detergent. It consists of sodium ion and lauryl sulphate anion.

When a detergent is applied on a cloth with a greasy stain, the detergent reduces the surface tension of the water due to which the surface of the cloth is wetted thoroughly. The lipophilic parts of the detergent anion will be soluble in grease and the hydrophilic parts will be soluble in water.
When the cloth is scrubbed, the grease is pulled away from the cloth and broken into smaller droplets. Repulsion between the droplets will cause them to be suspended in water, thus forming an emulsion. Thus, the droplets will not coagulate or redeposit on the cloth. As a result, rinsing will wash away the droplets making the cloth clean.
Hence, the correct option is A.
Note:
An advantage of detergent over soap is that detergents can be used even in hard water without any wastage but some of the soap gets wasted in hard water.
However, a disadvantage of detergents is that detergents having highly branched hydrocarbon chains cause pollution in rivers. This is because the side chains stop bacteria from attacking and breaking the chains which results in slow degradation of the detergent molecule leading to their accumulation in rivers. This causes water pollution.
Recently Updated Pages
JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

Isoelectronic Definition in Chemistry: Meaning, Examples & Trends

Ionisation Energy and Ionisation Potential Explained

Iodoform Reactions - Important Concepts and Tips for JEE

Introduction to Dimensions: Understanding the Basics

Instantaneous Velocity Explained: Formula, Examples & Graphs

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

Derivation of Equation of Trajectory Explained for Students

Understanding the Angle of Deviation in a Prism

Other Pages
JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

CBSE Notes Class 11 Chemistry Chapter 9 - Hydrocarbons - 2025-26

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

CBSE Notes Class 11 Chemistry Chapter 8 - Organic Chemistry Some Basic Principles And Techniques - 2025-26

JEE Advanced Weightage 2025 Chapter-Wise for Physics, Maths and Chemistry




