A ${C}_{8}{H}_{10}$ hydrocarbon is nitrated by $HN{O}_{3}$ and sulphuric acid. Two, and only two, ${C}_{8}{H}_{9}N{O}_{2}$ isomers are obtained. Which of the following fits into the evidence?
a) Ethyl benzene
b) Ortho-xylene
c) Meta-xylene
d) Para-xylene
Answer
261.6k+ views
Hint: Xylene is a common name for dimethyl benzene. The two methyl groups could be attached at ortho, meta or para position of the benzene ring.
Complete step by step solution:
● In the reaction given, if the only a single alkyl group is attached to the benzene ring, ethyl group in this case, the products that would be formed on reacting with $HN{O}_{3}$ and sulphuric acid will definitely have 3 isomers as the nitro group will have three carbons that it can attach to in the benzene ring. Those positions are ortho, meta, and para positions. But it is given in the question that only two isomers are formed of the reactant.
● If the attached group would have been a larger group, then the ortho position would have been blocked and only two isomers would have been formed. But this is not the case in this situation.
● For the product to have only two isomers it is important to have two alkyl groups attached to the benzene ring. And in this case, those alkyl groups are two identical methyl groups.
● But now the main question arises in which position these methyl groups are to be attached, ortho, meta or para.
● If they are attached at meta positions then, three isomers of the compound is possible.
● If they are attached at para position only one isomer is possible.
● But if we attach them at ortho position only two isomers are possible.
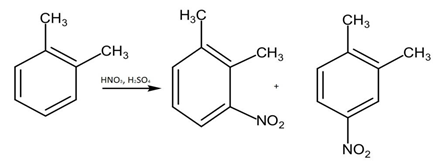
Hence, the ortho xylene will give us the desired product. Therefore, the correct answer is option (b).
Note: While making the isomers do make sure that you provide the numbering according to the standard rules and do not include the same isomers. If you don't follow the rules while making the isomers you might end up with more no. of isomers than expected.
Complete step by step solution:
● In the reaction given, if the only a single alkyl group is attached to the benzene ring, ethyl group in this case, the products that would be formed on reacting with $HN{O}_{3}$ and sulphuric acid will definitely have 3 isomers as the nitro group will have three carbons that it can attach to in the benzene ring. Those positions are ortho, meta, and para positions. But it is given in the question that only two isomers are formed of the reactant.
● If the attached group would have been a larger group, then the ortho position would have been blocked and only two isomers would have been formed. But this is not the case in this situation.
● For the product to have only two isomers it is important to have two alkyl groups attached to the benzene ring. And in this case, those alkyl groups are two identical methyl groups.
● But now the main question arises in which position these methyl groups are to be attached, ortho, meta or para.
● If they are attached at meta positions then, three isomers of the compound is possible.
● If they are attached at para position only one isomer is possible.
● But if we attach them at ortho position only two isomers are possible.

Hence, the ortho xylene will give us the desired product. Therefore, the correct answer is option (b).
Note: While making the isomers do make sure that you provide the numbering according to the standard rules and do not include the same isomers. If you don't follow the rules while making the isomers you might end up with more no. of isomers than expected.
Recently Updated Pages
Circuit Switching vs Packet Switching: Key Differences Explained

Dimensions of Pressure in Physics: Formula, Derivation & SI Unit

Matrices Explained: Definition, Types & Examples

Analytical Geometry: Concepts, Formulas & Problem Solving Guide

Gaseous and Liquid States Explained: Key Differences & Properties

Graphical Analysis of Kinematics: Concepts & Graphs Explained

Trending doubts
Understanding the Different Types of Solutions in Chemistry

JEE Main Marks vs Percentile 2026: Predict Your Score Easily

JEE Main Cutoff 2026: Category-wise Qualifying Percentile

JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main 2026 Session 2 Result Out Live Scorecard Link Active Cutoff Released

JEE Main Marks vs Rank 2026: Expected Rank for 300 to 0 Marks

Other Pages
Valentine Week 2026 Complete List, Dates, Quotes, Wishes & Celebration Guide

Mother's Day 2026: Date, History, Wishes, Quotes, Shayari, Gifts and Card Ideas

Ambedkar Jayanti 2026 – Date, History, Significance, Wishes, Quotes & Celebration

Navratri 2026: Day-to-Day Navratri Colours Guide with Correct Dates, Devi Names, and Significance

Labour Day 2026: Theme, History, Significance and Quotes

UP Board 10th Result 2026 OUT: Check Result, Pass Percentage, Toppers and Marksheet Download Steps




