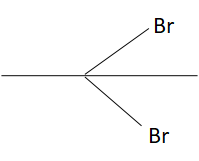
1-methyl ethylene oxide when treated with an excess of HBr produces:
A.
B.
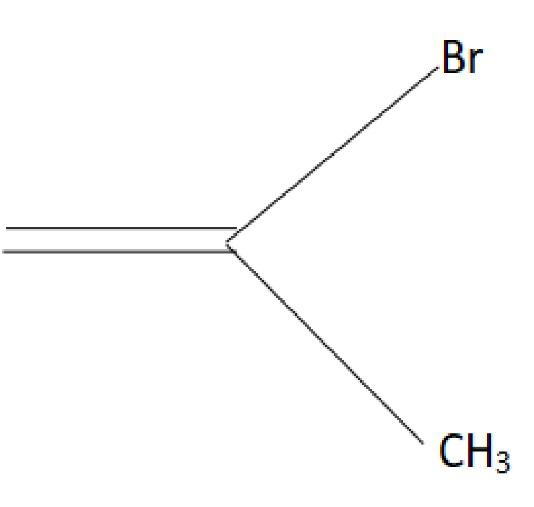
C.
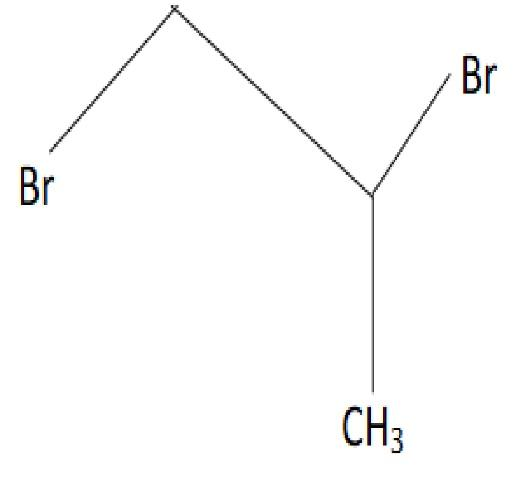
D.
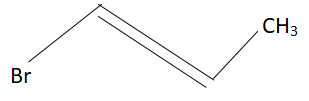
Answer
268.2k+ views
Hint: The three-membered ring epoxides can also be opened by anhydrous acids (like \[HBr\]) to form trans halohydrins. A halogen anion will attack the less substituted carbon in an \[{S_N}2\] like the reaction when both epoxide carbons are primary or secondary.
Complete Step by Step Solution:
1. 1-methyl ethylene oxide is a type of cyclic ether that reacts with strong acid to form a linear compound.
2. 1-methyl ethylene oxide undergoes a reaction with Hydrogen bromide in which a partial hydrogen ion attaches to the Oxygen atom with the lone pair and the Bromide ion is eliminated.
3. There are two possibilities to break this cyclic structure: one has only alpha Hydrogen and another has five alpha Hydrogen. So, the stability is higher for more alpha Hydrogen.
4. The negative charge Bromide ion attaches to the positive charge Carbon atom.
5. The excess Hydrogen Bromide reacts with the compound in which a positive charge Hydrogen atom makes a bond with the lone pair of an Oxygen atom and forms\[OH_2^ + \].
5. The negative charge Bromide ion attacks the positive position and forms 1,2-Dibromopropane with the elimination of water molecules.
6. The reaction of 1-methyl ethylene oxide with the excess Hydrogen bromide is expressed below:
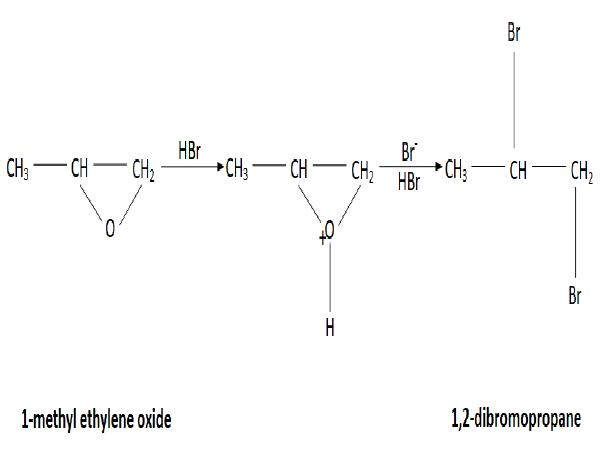
Image: Preparation of 1,2-dibromopropane
The correct option is (3).
Additional information:
The opening of epoxides under acidic conditions occurs in two ways:
1. The epoxide should be protonated first.
2. The nucleophilic attacks at the most substituted position.
Note: The three-membered ring is highly strained and readily open when it acts with strong acid under mild conditions.
Complete Step by Step Solution:
1. 1-methyl ethylene oxide is a type of cyclic ether that reacts with strong acid to form a linear compound.
2. 1-methyl ethylene oxide undergoes a reaction with Hydrogen bromide in which a partial hydrogen ion attaches to the Oxygen atom with the lone pair and the Bromide ion is eliminated.
3. There are two possibilities to break this cyclic structure: one has only alpha Hydrogen and another has five alpha Hydrogen. So, the stability is higher for more alpha Hydrogen.
4. The negative charge Bromide ion attaches to the positive charge Carbon atom.
5. The excess Hydrogen Bromide reacts with the compound in which a positive charge Hydrogen atom makes a bond with the lone pair of an Oxygen atom and forms\[OH_2^ + \].
5. The negative charge Bromide ion attacks the positive position and forms 1,2-Dibromopropane with the elimination of water molecules.
6. The reaction of 1-methyl ethylene oxide with the excess Hydrogen bromide is expressed below:

Image: Preparation of 1,2-dibromopropane
The correct option is (3).
Additional information:
The opening of epoxides under acidic conditions occurs in two ways:
1. The epoxide should be protonated first.
2. The nucleophilic attacks at the most substituted position.
Note: The three-membered ring is highly strained and readily open when it acts with strong acid under mild conditions.
Recently Updated Pages
Algebra Made Easy: Step-by-Step Guide for Students

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

JEE Energetics Important Concepts and Tips for Exam Preparation

Chemical Properties of Hydrogen - Important Concepts for JEE Exam Preparation

JEE General Topics in Chemistry Important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

How to Convert a Galvanometer into an Ammeter or Voltmeter

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

JEE Advanced 2026 Marks vs Rank: Estimate IIT Rank from Your Score

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

NCERT Solutions For Class 11 Chemistry In Hindi Chapter 1 Some Basic Concepts Of Chemistry - 2025-26

Understanding Electromagnetic Waves and Their Importance




