\[1,2\;\]di-bromocyclohexane on dehydrohalogenation gives
A. 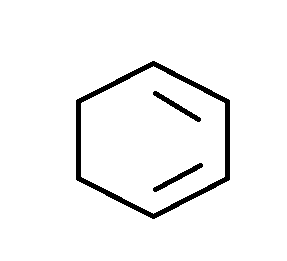
B. 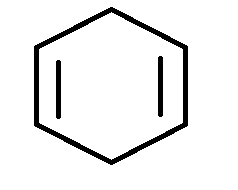
C. 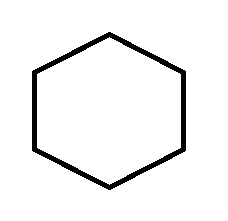
D. 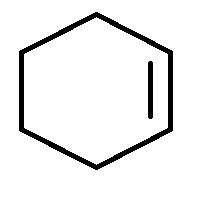
Answer
267.9k+ views
Hint: \[1,2\;\]di-bromocyclohexane has the following structure
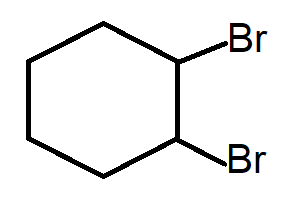
Dehydrohalogenation is a chemical reaction that undergoes removal of halogen and hydrogen to form alkene. ‘de’ means removal, ‘hydro’ means hydrogen, ‘halogen’ means halogen. The halogen and hydrogen must be present on adjacent positions.
Complete step-by-step answer:Dehydrohalogenation takes place in presence of a strong base like $KOH$. The $O{H^ - }$ accepts a hydrogen from the adjacent position of a good leaving group resulting in formation of positive charge which converts into a double bond by removal of the leaving group, namely halide for this particular reaction. In reality, the positive charge is actually not formed because the entire process occurs in a concerted manner. It is only to understand the basic mechanism. The elimination follows $E2$ mechanism which is a one step process with no intermediate (or positive charge) but only proceeds through the transition state. In \[1,2\;\] di-bromocyclohexane , the two bromides are present at \[1,2\;\] positions on cyclohexane. Each bromide will leave along with adjacent hydrogen to form a product similar to option A.
Hence A is the correct answer.
Option ‘A’ is correct
Additional Information: Alkene formation can follow Saytzeff elimination (more substituted alkene) or Hofmann elimination (less substituted alkene). Saytzeff elimination is followed in case of neutral molecules and Hofmann in case of charged species.
Note: Dehydrohalogenation requires proper stereo or proper orientation of the atoms. To undergo elimination, halogen and hydrogen must be anti-planner to each other. Hence, dehydrohalogenation is a stereospecific reaction. Dehalogenation reaction is also a type of reaction which eliminates two halogens present at \[1,2\;\]. But it is carried out only in presence of reagents like zinc dust,$NaI$, $MeOH$OR $EtOH$.

Dehydrohalogenation is a chemical reaction that undergoes removal of halogen and hydrogen to form alkene. ‘de’ means removal, ‘hydro’ means hydrogen, ‘halogen’ means halogen. The halogen and hydrogen must be present on adjacent positions.
Complete step-by-step answer:Dehydrohalogenation takes place in presence of a strong base like $KOH$. The $O{H^ - }$ accepts a hydrogen from the adjacent position of a good leaving group resulting in formation of positive charge which converts into a double bond by removal of the leaving group, namely halide for this particular reaction. In reality, the positive charge is actually not formed because the entire process occurs in a concerted manner. It is only to understand the basic mechanism. The elimination follows $E2$ mechanism which is a one step process with no intermediate (or positive charge) but only proceeds through the transition state. In \[1,2\;\] di-bromocyclohexane , the two bromides are present at \[1,2\;\] positions on cyclohexane. Each bromide will leave along with adjacent hydrogen to form a product similar to option A.
Hence A is the correct answer.
Option ‘A’ is correct
Additional Information: Alkene formation can follow Saytzeff elimination (more substituted alkene) or Hofmann elimination (less substituted alkene). Saytzeff elimination is followed in case of neutral molecules and Hofmann in case of charged species.
Note: Dehydrohalogenation requires proper stereo or proper orientation of the atoms. To undergo elimination, halogen and hydrogen must be anti-planner to each other. Hence, dehydrohalogenation is a stereospecific reaction. Dehalogenation reaction is also a type of reaction which eliminates two halogens present at \[1,2\;\]. But it is carried out only in presence of reagents like zinc dust,$NaI$, $MeOH$OR $EtOH$.
Recently Updated Pages
Algebra Made Easy: Step-by-Step Guide for Students

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

JEE Energetics Important Concepts and Tips for Exam Preparation

Chemical Properties of Hydrogen - Important Concepts for JEE Exam Preparation

JEE General Topics in Chemistry Important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
Understanding Electromagnetic Waves and Their Importance

Understanding the Different Types of Solutions in Chemistry

Electron Gain Enthalpy and Electron Affinity Explained

Degree of Dissociation: Meaning, Formula, Calculation & Uses

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Hybridisation in Chemistry – Concept, Types & Applications

Other Pages
CBSE Class 12 Chemistry Set 3 56/4/3 2025 Question Paper PDF & Answer Key

CBSE Class 12 Chemistry Question Paper Set 3 2025 with Answers

Test for Phenolic Group

CBSE Notes Class 12 Chemistry Chapter 10 - Biomolecules - 2025-26

CBSE Class 12 Chemistry Question Paper Set 2 (56/5/2) 2025 – PDF, Solutions & Analysis

Lactose is made up of A Galactose and glucose unit class 12 chemistry JEE_Main




