1, 3-Butadiene with bromine in molar ratio generates predominantly:
(A) 1,1-dibromo-2-butene
(B) 1,4-dibromo-2-butene
(C) 1,2-dibromo-2-butene
(D) 1,3-dibromo-2-butene
Answer
265.8k+ views
Hint: First draw the structure of the molecule and then form the product by keeping in mind the conjugation due to the 2 double bonds present.
Complete step by step answer:
-Let us talk about the structure of 1,3-butadiene. It is a 4 carbon organic molecule with double bonds at 1 and 3 positions. It’s molecular formula is: ${C_4}{H_6}$.
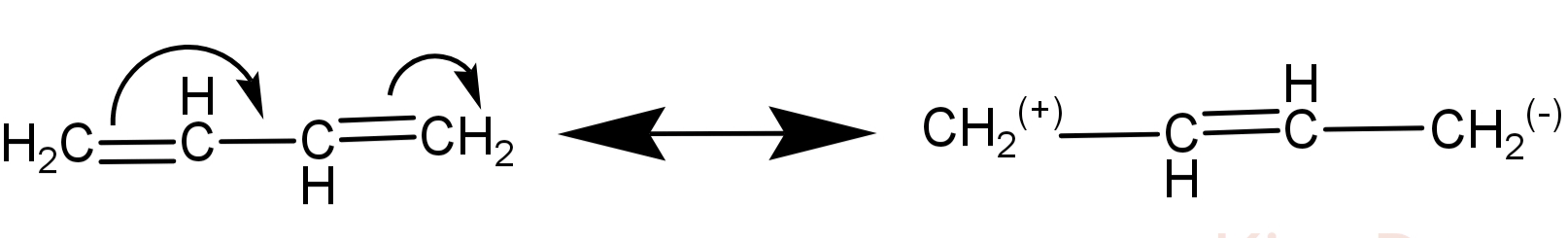
The 2 double bonds make 1,3-butadiene structure to be conjugated (which means there is delocalisation of electrons in this structure). Its conjugated structures are as follows:
- When halogen attacks on a double bond there is scope for 2 types of products:
One which is formed by 1,2-addition and is kinetically favoured product (because it is formed fast)
And the other is formed by 1,4-addition and is a thermodynamically favoured product because it is associated with thermodynamic stability.
This thermodynamic stability gives a reason for the formation of a 1,4-addition product.
-Also when any halogen attacks on a double bond, there is addition of that halogen on that double bond and a 1,2-addition product might be formed. But this occurs only if there is no conjugation in that double bond.
Here in 1,3-butadiene there is conjugation of double bond and so addition of bromine is 1,4-addition. This means that the double bond shifts between the 2nd and the 3rd carbon (conjugated structure) and Br atoms are added at 1 and 4 position.
The above given reasons favour the formation of a 1,4-addition product. Thus the product formed is 1,4-butadiene.
This is shown in the following reaction:
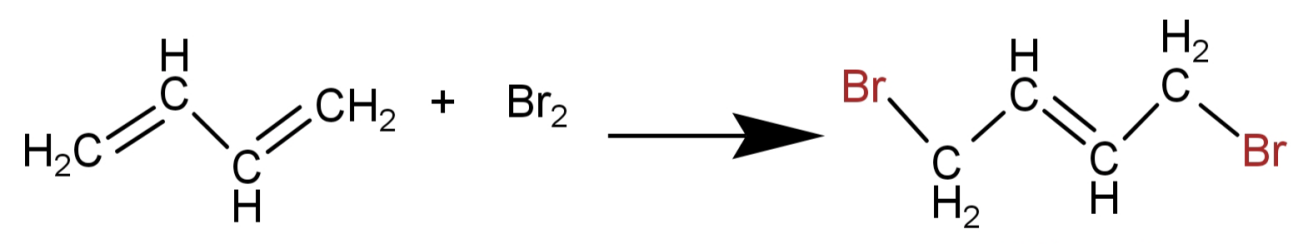
So, the correct option is: (B)1,4-dibromo-2-butene.
Note: Most common mistake we make here is of doing 1,2-addition which means adding 2 Br atoms at 1 and 2 positions of the molecule.
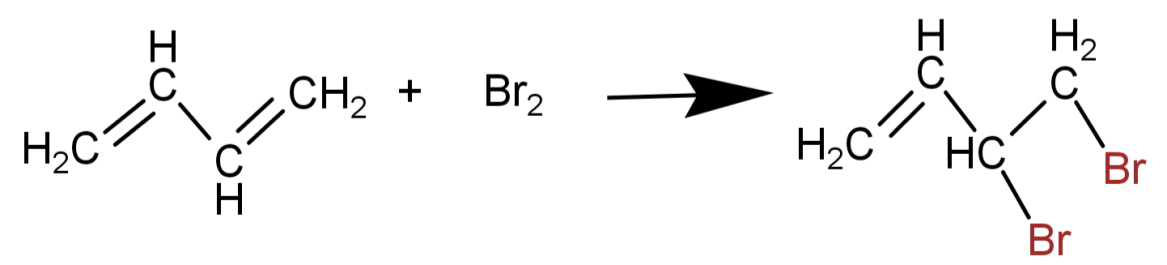
But this is wrong because conjugation is happening here. So, remember that when there is conjugation due to a double bond there will be 1,4- addition.
Complete step by step answer:
-Let us talk about the structure of 1,3-butadiene. It is a 4 carbon organic molecule with double bonds at 1 and 3 positions. It’s molecular formula is: ${C_4}{H_6}$.
The 2 double bonds make 1,3-butadiene structure to be conjugated (which means there is delocalisation of electrons in this structure). Its conjugated structures are as follows:

- When halogen attacks on a double bond there is scope for 2 types of products:
One which is formed by 1,2-addition and is kinetically favoured product (because it is formed fast)
And the other is formed by 1,4-addition and is a thermodynamically favoured product because it is associated with thermodynamic stability.
This thermodynamic stability gives a reason for the formation of a 1,4-addition product.
-Also when any halogen attacks on a double bond, there is addition of that halogen on that double bond and a 1,2-addition product might be formed. But this occurs only if there is no conjugation in that double bond.
Here in 1,3-butadiene there is conjugation of double bond and so addition of bromine is 1,4-addition. This means that the double bond shifts between the 2nd and the 3rd carbon (conjugated structure) and Br atoms are added at 1 and 4 position.
The above given reasons favour the formation of a 1,4-addition product. Thus the product formed is 1,4-butadiene.
This is shown in the following reaction:

So, the correct option is: (B)1,4-dibromo-2-butene.
Note: Most common mistake we make here is of doing 1,2-addition which means adding 2 Br atoms at 1 and 2 positions of the molecule.

But this is wrong because conjugation is happening here. So, remember that when there is conjugation due to a double bond there will be 1,4- addition.
Recently Updated Pages
States of Matter Chapter For JEE Main Chemistry

Types of Solutions in Chemistry: Explained Simply

Difference Between Crystalline and Amorphous Solid: Table & Examples

Know The Difference Between Fluid And Liquid

[Awaiting the three content sources: Ask AI Response, Competitor 1 Content, and Competitor 2 Content. Please provide those to continue with the analysis and optimization.]

Sign up for JEE Main 2026 Live Classes - Vedantu

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

JEE Advanced 2026 Marks vs Rank: Estimate IIT Rank from Your Score

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

NCERT Solutions For Class 11 Chemistry In Hindi Chapter 1 Some Basic Concepts Of Chemistry - 2025-26

How to Convert a Galvanometer into an Ammeter or Voltmeter




