What is Markovnikov’s rule? Explain with an example.
Answer
561.7k+ views
Hint: We should know that Markovnikov’s rule is an empirical rule which is used to predict regioselectivity of electrophilic addition reaction of alkenes and alkynes. Keeping the purpose in mind, we need to describe the mechanism of Markovnikov's rule.
Complete step by step solution:
The Markovnikov’s rule states that with the addition of a protic acid HX to an asymmetric alkene, the acid hydrogen (H) gets attached to the carbon with more hydrogen substituents, and the halide (X) group gets attached to the carbon with more alkyl substituents.
Let us explain Markovnikov's rule with the help of a simple example.
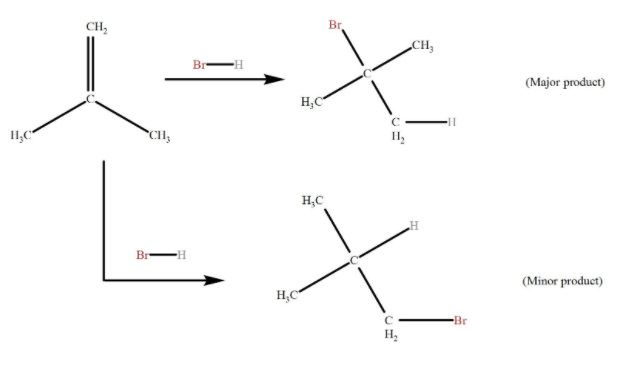
When a protic acid HC (X = Cl, Br, I) is added to an asymmetrically substituted alkene, addition of acidic hydrogen takes place at the less substituted carbon atom of the double bond, while X is added to the more alkyl substituted carbon atom. In other words, hydrogen is added to the carbon atom with more number of hydrogen atoms attached to it and halide is added to the carbon with the least number of hydrogen atoms.
We should know that the driving force behind the reaction is the formation of carbocation in addition of H to the alkene, in the first step of the reaction. The most stable carbocation is formed when H is added to carbon having more number of hydrogen atoms already attached, due to factors like induction and hyperconjugation, which gives the major product with Br added to less hydrogen rich carbon.
Less stable carbocation (H added to less hydrogen rich carbon) is also present in small quantities, forming minor products in addition to Br to carbon having more hydrogen attached.
Note: There are various practical applications of Markovnikov’s rule. Some of them are Halohydrin formation (in alcohol and water), Oxymercurationand Demercuration and Acid catalysed hydration. These reactions are the laboratory usages of Markovnikov’s rule and are used in a variety of chemical processes.
Complete step by step solution:
The Markovnikov’s rule states that with the addition of a protic acid HX to an asymmetric alkene, the acid hydrogen (H) gets attached to the carbon with more hydrogen substituents, and the halide (X) group gets attached to the carbon with more alkyl substituents.
Let us explain Markovnikov's rule with the help of a simple example.
When a protic acid HC (X = Cl, Br, I) is added to an asymmetrically substituted alkene, addition of acidic hydrogen takes place at the less substituted carbon atom of the double bond, while X is added to the more alkyl substituted carbon atom. In other words, hydrogen is added to the carbon atom with more number of hydrogen atoms attached to it and halide is added to the carbon with the least number of hydrogen atoms.
We should know that the driving force behind the reaction is the formation of carbocation in addition of H to the alkene, in the first step of the reaction. The most stable carbocation is formed when H is added to carbon having more number of hydrogen atoms already attached, due to factors like induction and hyperconjugation, which gives the major product with Br added to less hydrogen rich carbon.
Less stable carbocation (H added to less hydrogen rich carbon) is also present in small quantities, forming minor products in addition to Br to carbon having more hydrogen attached.

Note: There are various practical applications of Markovnikov’s rule. Some of them are Halohydrin formation (in alcohol and water), Oxymercurationand Demercuration and Acid catalysed hydration. These reactions are the laboratory usages of Markovnikov’s rule and are used in a variety of chemical processes.
Recently Updated Pages
Disproportionation Reaction: Definition, Example & JEE Guide

Hess Law of Constant Heat Summation: Definition, Formula & Applications

JEE General Topics in Chemistry Important Concepts and Tips

JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

JEE Advanced 2026 Marks vs Rank: Estimate IIT Rank from Your Score

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

Understanding the Different Types of Solutions in Chemistry

NCERT Solutions For Class 11 Chemistry In Hindi Chapter 1 Some Basic Concepts Of Chemistry - 2025-26




