



Understanding Simple Diffusion and How Substances Move Across Membranes
Simple diffusion is a fundamental biological process in which molecules move from a region of higher concentration to a region of lower concentration until equilibrium is achieved. This movement happens naturally and does not require any external energy (ATP).
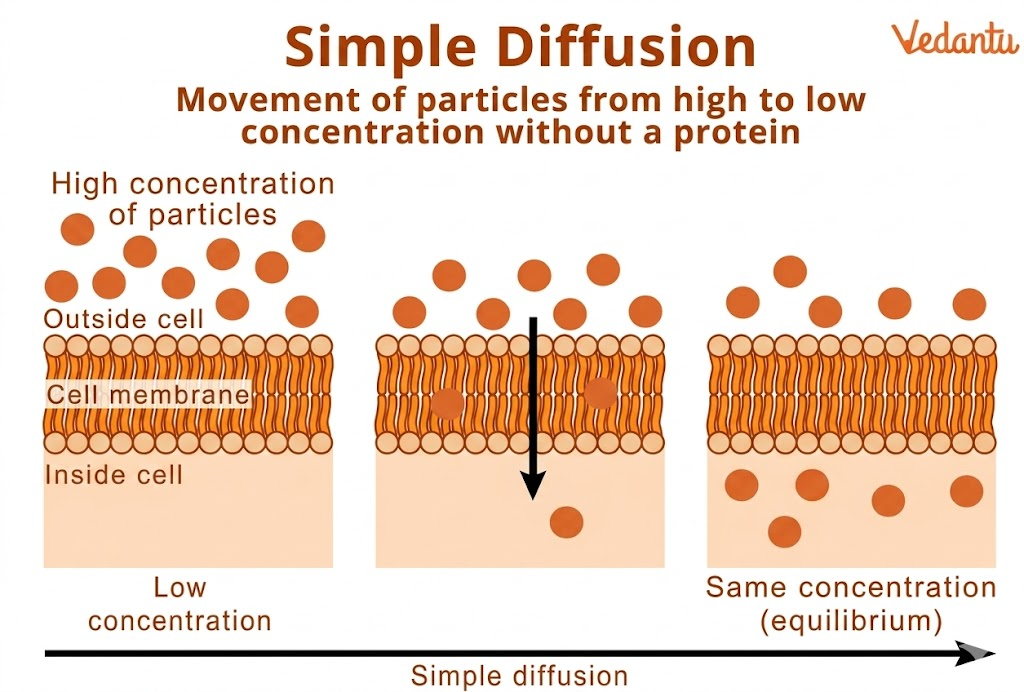
This process is driven by the natural tendency of molecules to spread evenly in available space. The concentration gradient itself acts as a form of stored energy that powers the movement of particles.
Simple diffusion is commonly seen in biological systems where small and non-polar molecules, such as oxygen and carbon dioxide, pass directly through the cell membrane.
Key Features of Simple Diffusion
Movement from high concentration to low concentration
No energy (ATP) required
Occurs along a concentration gradient
Does not require carrier or channel proteins
Continues until equilibrium is reached
Mostly involves small, non-polar molecules
Mechanism of Simple Diffusion
The mechanism of simple diffusion is based on random molecular motion and concentration differences.
1. Random Movement of Molecules
Molecules are in constant motion due to their kinetic energy. They collide with each other continuously, a process known as pedesis.
2. Movement Along Concentration Gradient
In regions where concentration is high, molecules are crowded and collide more often. They tend to move toward areas with more space (lower concentration).
3. Role of Membrane
If the molecules are small enough, they pass directly through the phospholipid bilayer of the membrane without assistance.
4. Achievement of Equilibrium
Diffusion continues until the concentration becomes equal on both sides. At this point, there is no net movement of molecules.
Simple Diffusion Example
A clear example of simple diffusion helps in understanding the concept effectively.
Example 1: Gas Exchange in Cells
Oxygen moves from areas of high concentration (outside the cell) to lower concentration (inside the cell). Similarly, carbon dioxide diffuses out of the cell.
Example 2: Diffusion in Bacteria
Bacteria rely heavily on diffusion because they lack complex transport systems. They absorb oxygen, water, and small nutrients through simple diffusion.
Example 3: Perfume Spreading in Air
When perfume is sprayed in one corner of a room, its molecules gradually spread throughout the room due to diffusion.
Factors Affecting Simple Diffusion
The factors affecting simple diffusion determine how fast or slow the diffusion process occurs.
1. Size of Molecules
Smaller molecules diffuse faster, while larger molecules face more resistance.
2. Temperature
Higher temperature increases kinetic energy, leading to faster diffusion.
3. Concentration Gradient
A steeper gradient (larger difference in concentration) increases the rate of diffusion.
4. Nature of the Membrane
Permeability of the membrane affects diffusion. Non-polar molecules pass easily, while polar molecules cannot.
5. Medium of Diffusion
Diffusion occurs faster in gases than in liquids due to less resistance.
Simple Diffusion vs Other Transport Mechanisms
Understanding simple diffusion becomes easier when compared with other transport processes.
1. Simple Diffusion vs Facilitated Diffusion
Simple diffusion: No proteins required
Facilitated diffusion: Requires carrier or channel proteins
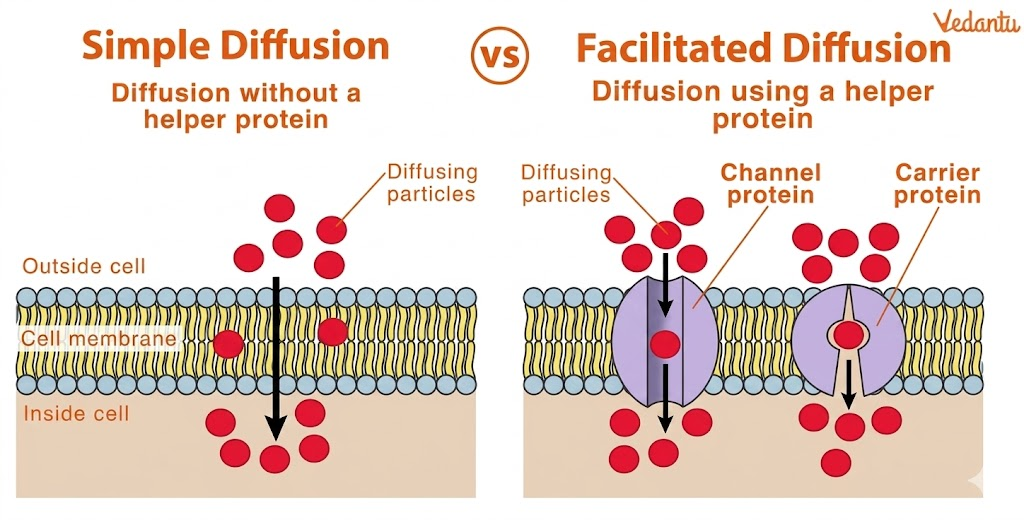
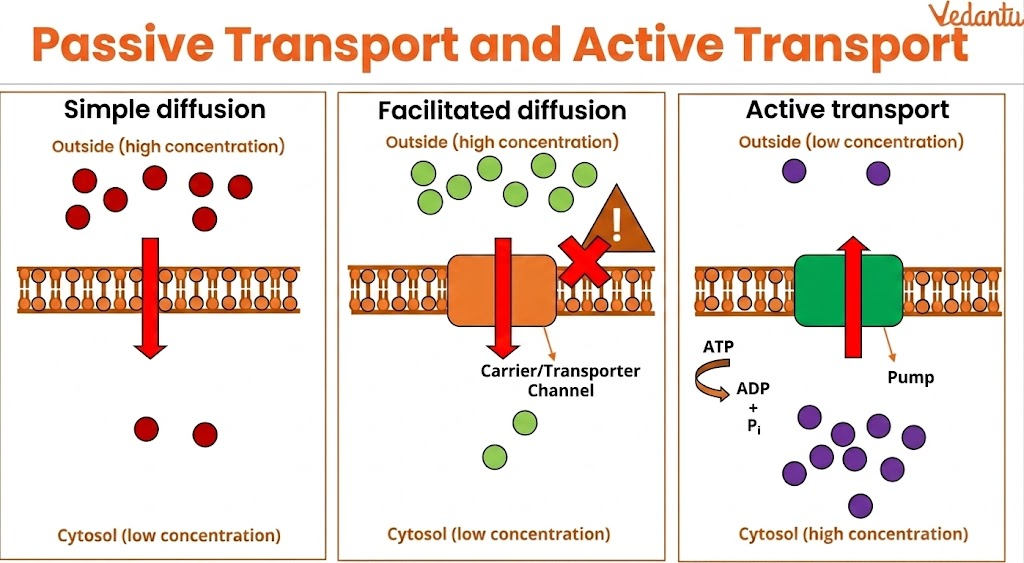
2. Simple Diffusion vs Active Transport
Simple diffusion: Passive, no energy needed
Active transport: Requires energy and moves against gradient
Role of Simple Diffusion in Living Organisms
Simple diffusion plays a critical role in maintaining life processes:
Gas exchange in lungs and tissues
Movement of small nutrients into cells
Removal of waste products
Maintaining cellular balance
Even though it is a simple process, it is essential for survival, especially in unicellular organisms.
Important Concept: Diffusion and Osmosis
Water also moves through membranes by diffusion, a process known as osmosis.
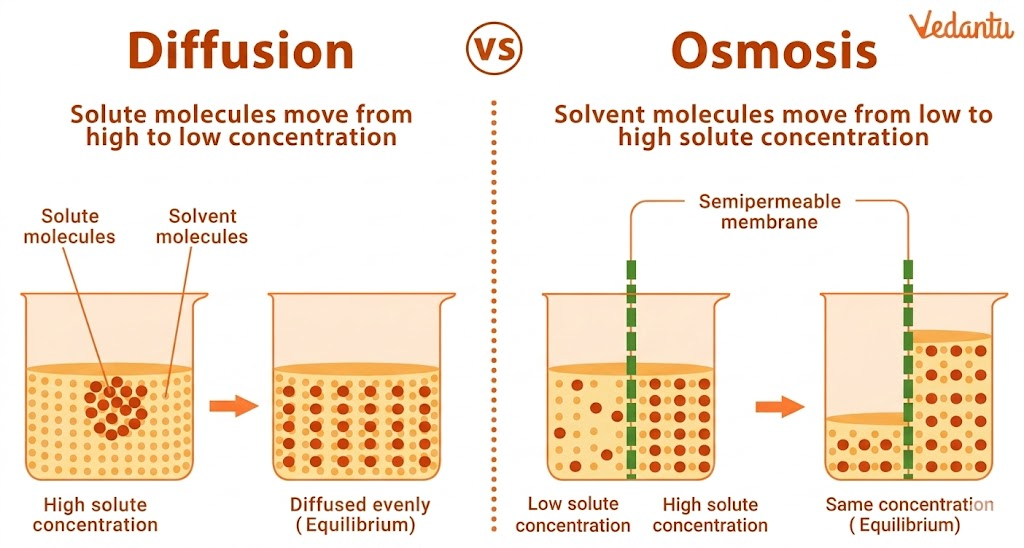
Osmosis is a special case of diffusion where solvent molecules move across a semipermeable membrane from higher to lower water potential.
Quick Revision Table
Key Takeaways
Simple diffusion is a passive process driven by concentration gradient
No ATP or transport proteins are required
It continues until equilibrium is reached
It is essential for gas exchange and basic cellular functions
Factors like temperature, size, and gradient affect its rate
In this article, we explored Simple Diffusion, its key processes, real-life significance, comparison with similar processes, and how to solve NEET-related questions. To learn more and build confidence, keep practicing with Vedantu.
Facilitated Diffusion – Understand MCQ differences and prevent confusion.
Cell Membrane – Explore membrane structure and transport roles.
Diffusion Means of Transport – See types of diffusion in biology.
Passive Transport – Compare all passive processes for NEET’s “no ATP” questions.
Cell Structure and Function – Review broader cell concepts related to diffusion.
Osmosis – Learn water movement and avoid mistakes with diffusion.
MCQs on Animal Cell – Test your knowledge with NEET-style questions.
Life Processes – Connect diffusion with whole-organism energy flow.
Cell Transport and Its Types – Understand transport diversity for application-based questions.
Difference Between Active and Passive Transport – Strengthen key NEET comparison skills.
FAQs on Simple Diffusion: Definition, Diagram, Mechanism, Examples and Factors Affecting It
1. What is meant by simple diffusion?
Simple diffusion is the passive movement of molecules from a region of higher concentration to lower concentration without using energy or transport proteins.
2. What is simple and facilitated diffusion?
Both are passive transport processes.
Simple diffusion: Molecules move directly through the membrane
Facilitated diffusion: Molecules move through the membrane with the help of carrier or channel proteins
3. Why is simple diffusion important?
Simple diffusion is important because it helps cells:
take in useful substances like oxygen
remove waste products like carbon dioxide
maintain internal balance without using energy
4. What are 5 examples of diffusion?
Five common examples of diffusion are:
smell of perfume spreading in a room
food colouring spreading in water
tea spreading in hot water
oxygen moving from lungs into blood
smell of cooking spreading through air
5. What factors affect simple diffusion?
The main factors affecting simple diffusion are:
temperature
concentration gradient
surface area
size of molecules
nature of the membrane or medium
6. Which diffusion is faster?
Diffusion is fastest in gases and slowest in solids because gas particles move more freely.
7. What are the two types of diffusion?
The two main types of diffusion are:
Simple diffusion
Facilitated diffusion
8. What is the difference between diffusion and simple diffusion?
Diffusion is the general movement of particles from high concentration to low concentration.
9. Can CO2 pass through simple diffusion?
Yes, CO2 can pass through simple diffusion because it is a small molecule and can move directly across the cell membrane.
10. What molecules use simple diffusion?
Molecules that use simple diffusion are usually small and non-polar, such as:
oxygen (O₂)
carbon dioxide (CO₂)
some lipid-soluble molecules























