Which one of the following octahedral complexes will not show geometric isomerism (A and B are monodentate ligands)
A) $\left[ M{{A}_{5}}B \right]$
B) $\left[ M{{A}_{2}}{{B}_{4}} \right]$
C) $\left[ M{{A}_{3}}{{B}_{3}} \right]$
D) $\left[ M{{A}_{4}}{{B}_{2}} \right]$
Answer
264.6k+ views
Hint: The geometrical isomerism is only exhibited by the square planar complexes and the octahedral complexes due to the presence of planar geometry. The only condition required by a coordination complex to show geometrical isomerism is the positioning of the ligands should not be equivalent i.e., the structures should not superimpose to each other.
Complete answer:In the octahedral complexes which are heteroleptic in nature i.e., the complexes in which the metal ion is surrounded by more than one type of ligands, show geometrical isomerism due to the possibility of the ligands to get arranged in different positions.
Geometrical isomerism is broadly categorized into two types i.e., cis-geometrical isomerism and trans-geometrical isomerism. If the identical ligands are placed in the adjacent position with respect to the central metal ion then it is said to be cis-geometrical isomerism. If the identical ligands are placed in the opposite direction, then it is called trans-geometrical isomerism.
In the case of octahedral complexes having monodentate ligands, the geometrical isomerism can be summarised as follows:
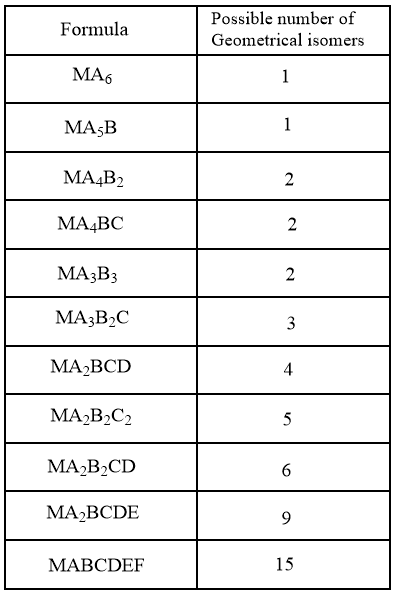
Where, M is the central metal ion and A, B, C, D, E and F are the monodentate ligands.
As summarised in the data, it is observed that the complexes of type ${{\left[ M{{A}_{6}} \right]}^{n\pm }}$ and ${{\left[ M{{A}_{5}}B \right]}^{n\pm }}$ does not show geometrical isomerism because all the corners of the regular octahedron are equivalent and no arrangement is observed. An example of such complexes is given below:
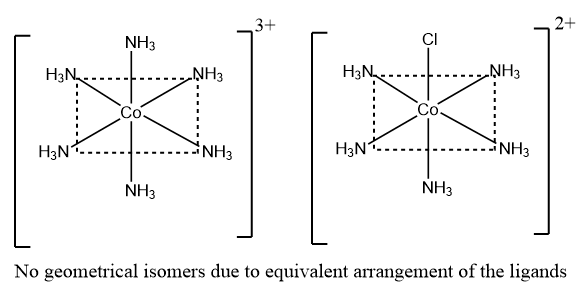
Thus, among the given options, the complex which does not show geometrical isomerism is $\left[ M{{A}_{5}}B \right]$, and therefore option A is the correct answer.
Note: It is important to note that cis geometrical isomerism is also known as facial or fac-isomer as the similar groups are occupied in the adjacent positions of the octahedral faces while the trans geometrical isomerism is known as meridional or mer-isomer because the position of the donor atom is around the meridian of the octahedron.
Complete answer:In the octahedral complexes which are heteroleptic in nature i.e., the complexes in which the metal ion is surrounded by more than one type of ligands, show geometrical isomerism due to the possibility of the ligands to get arranged in different positions.
Geometrical isomerism is broadly categorized into two types i.e., cis-geometrical isomerism and trans-geometrical isomerism. If the identical ligands are placed in the adjacent position with respect to the central metal ion then it is said to be cis-geometrical isomerism. If the identical ligands are placed in the opposite direction, then it is called trans-geometrical isomerism.
In the case of octahedral complexes having monodentate ligands, the geometrical isomerism can be summarised as follows:

Where, M is the central metal ion and A, B, C, D, E and F are the monodentate ligands.
As summarised in the data, it is observed that the complexes of type ${{\left[ M{{A}_{6}} \right]}^{n\pm }}$ and ${{\left[ M{{A}_{5}}B \right]}^{n\pm }}$ does not show geometrical isomerism because all the corners of the regular octahedron are equivalent and no arrangement is observed. An example of such complexes is given below:

Thus, among the given options, the complex which does not show geometrical isomerism is $\left[ M{{A}_{5}}B \right]$, and therefore option A is the correct answer.
Note: It is important to note that cis geometrical isomerism is also known as facial or fac-isomer as the similar groups are occupied in the adjacent positions of the octahedral faces while the trans geometrical isomerism is known as meridional or mer-isomer because the position of the donor atom is around the meridian of the octahedron.
Recently Updated Pages
JEE Main Mock Test 2025-26: Principles Related To Practical

JEE Main 2025-26 Experimental Skills Mock Test – Free Practice

JEE Main 2025-26 Electronic Devices Mock Test: Free Practice Online

JEE Main 2025-26 Mock Tests: Free Practice Papers & Solutions

JEE Main 2025-26: Magnetic Effects of Current & Magnetism Mock Test

JEE Main Statistics and Probability Mock Test 2025-26

Trending doubts
JEE Main Marks vs Percentile 2026: Predict Your Score Easily

JEE Main Cutoff 2026: Category-wise Qualifying Percentile

JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Marks vs Rank 2026: Expected Rank for 300 to 0 Marks

NIT Cutoff 2026: Tier-Wise Opening and Closing Ranks for B.Tech. Admission

JEE Mains 2026 Subject Wise Percentile Explained

Other Pages
Valentine Week 2026 Complete List, Dates, Quotes, Wishes & Celebration Guide

Mother's Day 2026: Date, History, Wishes, Quotes, Shayari, Gifts and Card Ideas

Navratri 2026: Day-to-Day Navratri Colours Guide with Correct Dates, Devi Names, and Significance

UP Board 10th Result 2026 OUT: Check Result, Pass Percentage, Toppers and Marksheet Download Steps

Odisha 10th Result 2026 (LIVE Soon): BSE HSC Result Link, Date & Updates

Chaitra Navratri 2026 Calendar Dates, Ghatsthapana Shuabh Muhurat Timings, Rituals, and Significance





