Which of the following is a meso compound?
A. 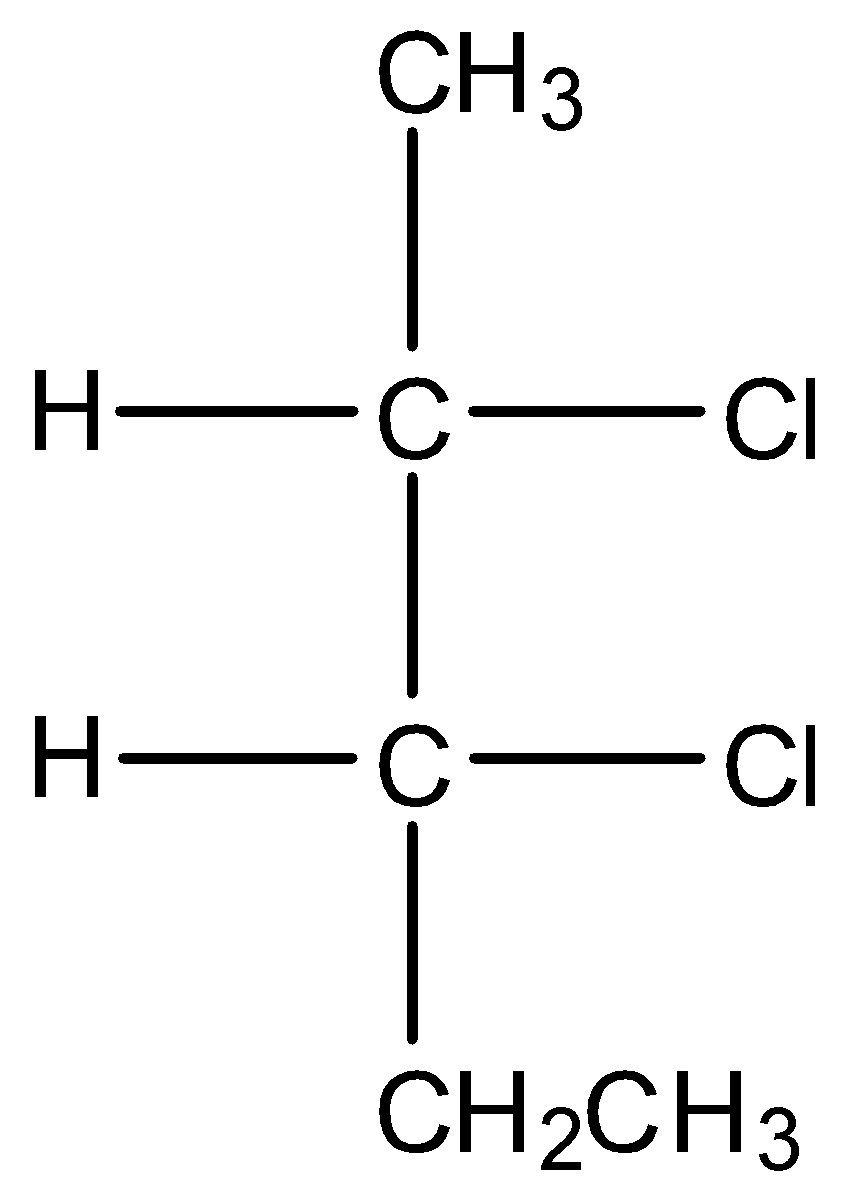
B. 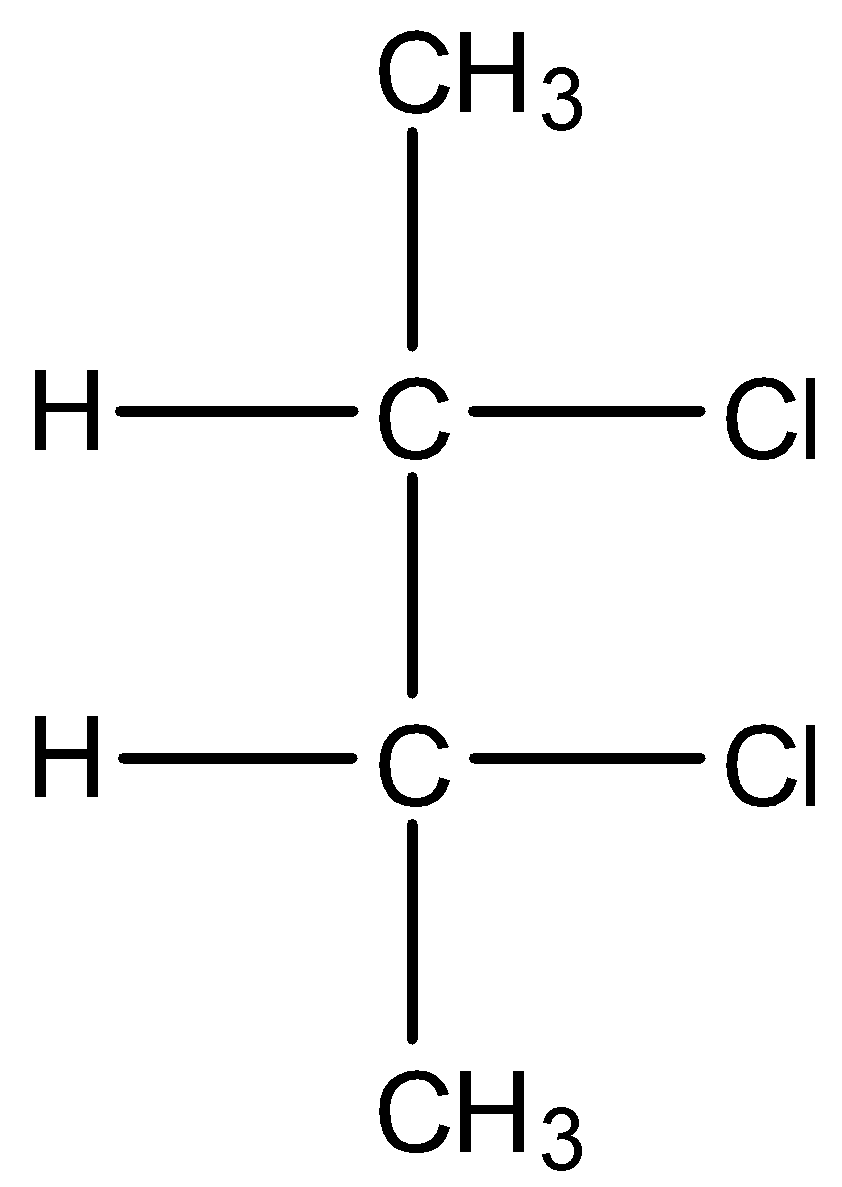
C. 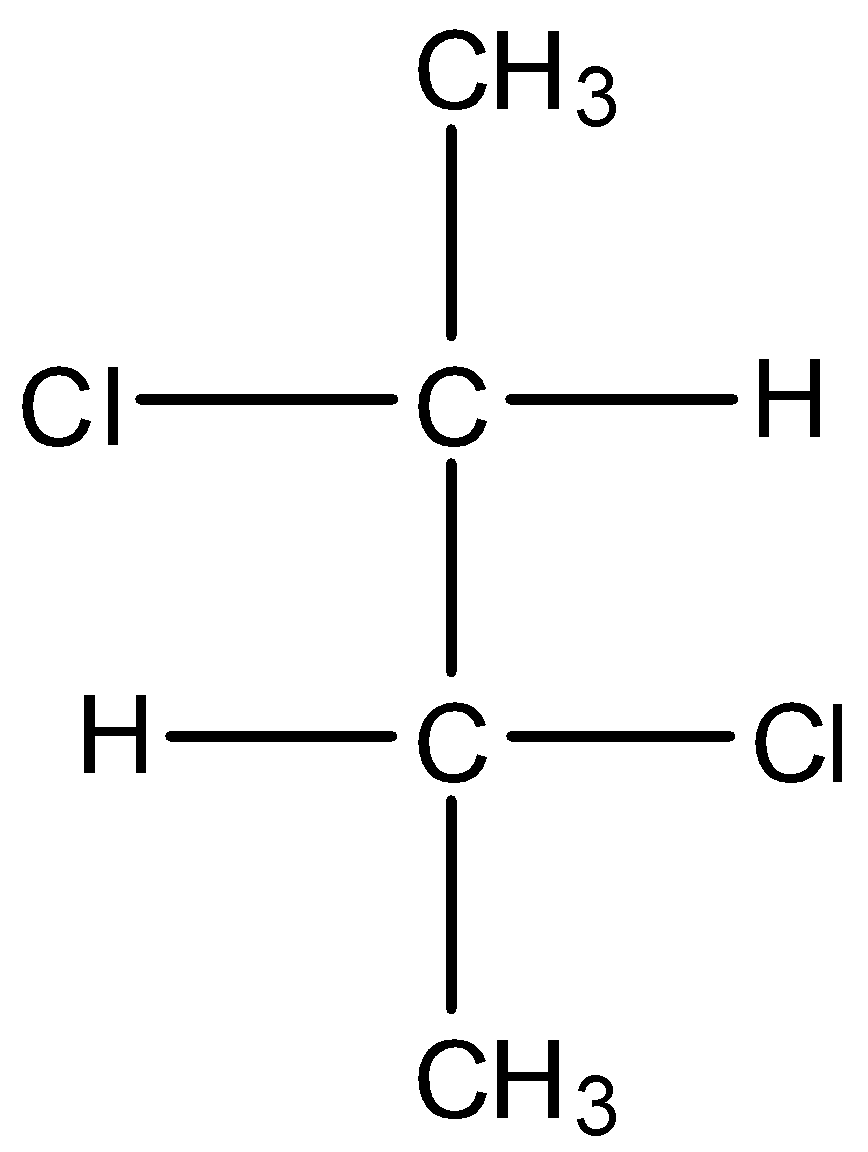
D. 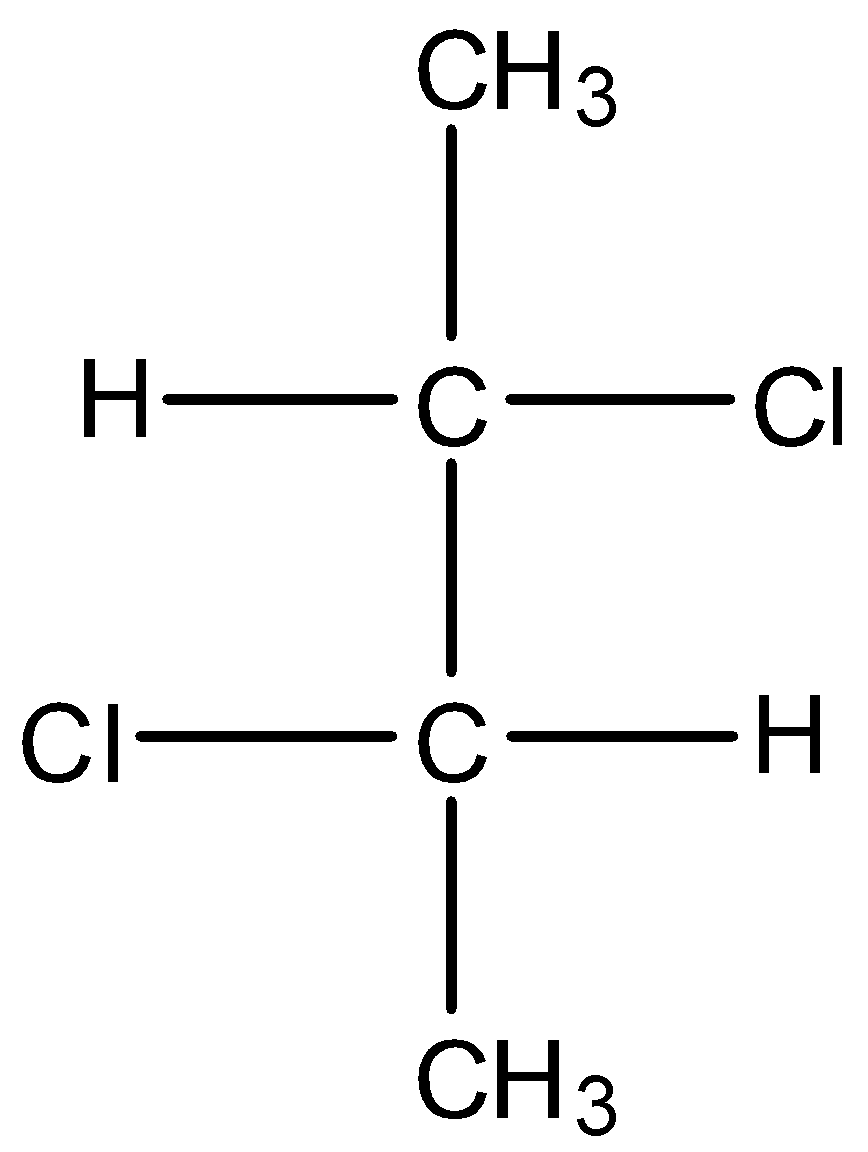
Answer
257.4k+ views
Hint: “A meso compound should contain two or more identical substituted stereo centers. Also, it has an internal symmetry plane that divides the compound in half. These two halves reflect each other by the internal mirror”.
Complete step by step answer:
* A meso compound should possess two or more substituted stereo centers and the compound should have the property of internal plane of symmetry means an imaginary plane should divide the compounds into two halves of identical structures nothing but mirror images.
* Coming to option A, the compound has two stereo centers but does not possess the property of internal plane of symmetry.

* Coming to option B, the compound has two stereocenters and an imaginary plane dividing the molecule into two halves which are mirror images. So, option B is the correct answer.

* Coming to option C, the compound has two stereo centers but does not possess the property of internal plane of symmetry.

* Coming to option D, the compound has two stereo centers but does not possess the property of internal plane of symmetry.

* So, the correct answer is B.
Note: Don’t be confused with meso, stereo center and internal plane of symmetry.
All compounds that have stereocenters won’t be meso compounds, the compounds which have more than two stereocenters and internal plane of symmetry are called meso compounds.
Complete step by step answer:
* A meso compound should possess two or more substituted stereo centers and the compound should have the property of internal plane of symmetry means an imaginary plane should divide the compounds into two halves of identical structures nothing but mirror images.
* Coming to option A, the compound has two stereo centers but does not possess the property of internal plane of symmetry.

* Coming to option B, the compound has two stereocenters and an imaginary plane dividing the molecule into two halves which are mirror images. So, option B is the correct answer.

* Coming to option C, the compound has two stereo centers but does not possess the property of internal plane of symmetry.

* Coming to option D, the compound has two stereo centers but does not possess the property of internal plane of symmetry.

* So, the correct answer is B.
Note: Don’t be confused with meso, stereo center and internal plane of symmetry.
All compounds that have stereocenters won’t be meso compounds, the compounds which have more than two stereocenters and internal plane of symmetry are called meso compounds.
Recently Updated Pages
JEE Main Mock Test 2025-26: Principles Related To Practical

JEE Main 2025-26 Mock Test: Organic Compounds Containing Nitrogen

JEE Main Mock Test 2025-26: Purification & Characterisation of Organic Compounds

JEE Main 2025-26 Mock Test: Principles Related To Practical

JEE Main Mock Test 2025-26: Principles & Best Practices

Purification and Characterisation of Organic Compounds JEE Main 2025-26 Mock Test

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

JEE Main Colleges 2026: Complete List of Participating Institutes

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Hybridisation in Chemistry – Concept, Types & Applications

Other Pages
JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 10 Biomolecules - 2025-26

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26




