Which of the following compounds doesn't have a linear structure
A. Carbon dioxide
B. Sulfur dioxide
C. Beryllium chloride
D. Ethylene
Answer
249k+ views
Hint: Linear compounds undergo sp hybridization and remain at an angle of \[180^o\] with each other. When multiple bonds are involved in a molecule, only one electron group is considered for multiple bonds for hybridization in these types of compounds.
Complete step by step solution:A. Carbon dioxide
Carbon is the central atom. It has four valence electrons. Each oxygen atom is doubly bonded to the carbon atom.
There are two sigma bonds and two pi-bonds on C.
So, there will be sp hybridization.
So, it has a linear structure.
So, A is incorrect.
B. Sulfur dioxide
Sulfur is the central atom. It has six electrons in its valence shell.
Each oxygen atom is doubly bonded to the carbon atom.
There are two bond pairs and one lone pair on S.
So, there will be \[sp^2\] hybridization.
So, it has a bent structure.
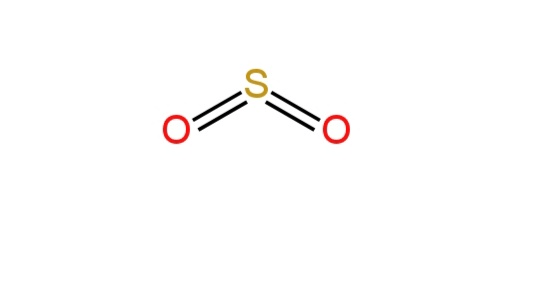
Structure: Structure of sulfur dioxide
So, B is correct.
C. Beryllium chloride
Be is the central atom. It has two valence electrons.
Each chlorine atom is bonded to the Be atom through a sigma bond.
There are two bond pairs.
So, there will be sp hybridization.
So, it has a linear structure.
So, C is incorrect.
D. Ethylene
In this, each carbon atom is bound with one hydrogen atom and the two carbon atoms are joined together. The bond between the carbon and carbon atom is a triple bond.
Considering the triple bond as one electron group, we see that there are two electron groups on carbon.
Here the hybridization will be sp.
The structure of ethylene is provided below:
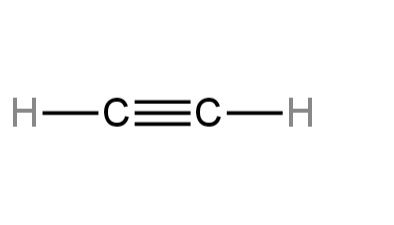
So, D is incorrect.
Sulfur dioxide has a bent structure.
So, option B is correct.
Note: Sulfur dioxide is utilized in numerous industries. It's utilized to manufacture sulfuric acid, paper, and food preservatives. It works as a bleaching agent to pull out the excess chlorine and as a disinfectant. In a cold storage plant, it behaves as a refrigerant.
Complete step by step solution:A. Carbon dioxide
Carbon is the central atom. It has four valence electrons. Each oxygen atom is doubly bonded to the carbon atom.
There are two sigma bonds and two pi-bonds on C.
So, there will be sp hybridization.
So, it has a linear structure.
So, A is incorrect.
B. Sulfur dioxide
Sulfur is the central atom. It has six electrons in its valence shell.
Each oxygen atom is doubly bonded to the carbon atom.
There are two bond pairs and one lone pair on S.
So, there will be \[sp^2\] hybridization.
So, it has a bent structure.

Structure: Structure of sulfur dioxide
So, B is correct.
C. Beryllium chloride
Be is the central atom. It has two valence electrons.
Each chlorine atom is bonded to the Be atom through a sigma bond.
There are two bond pairs.
So, there will be sp hybridization.
So, it has a linear structure.
So, C is incorrect.
D. Ethylene
In this, each carbon atom is bound with one hydrogen atom and the two carbon atoms are joined together. The bond between the carbon and carbon atom is a triple bond.
Considering the triple bond as one electron group, we see that there are two electron groups on carbon.
Here the hybridization will be sp.
The structure of ethylene is provided below:

So, D is incorrect.
Sulfur dioxide has a bent structure.
So, option B is correct.
Note: Sulfur dioxide is utilized in numerous industries. It's utilized to manufacture sulfuric acid, paper, and food preservatives. It works as a bleaching agent to pull out the excess chlorine and as a disinfectant. In a cold storage plant, it behaves as a refrigerant.
Recently Updated Pages
JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

Isoelectronic Definition in Chemistry: Meaning, Examples & Trends

Ionisation Energy and Ionisation Potential Explained

Iodoform Reactions - Important Concepts and Tips for JEE

Introduction to Dimensions: Understanding the Basics

Instantaneous Velocity Explained: Formula, Examples & Graphs

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

Derivation of Equation of Trajectory Explained for Students

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Other Pages
JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

CBSE Notes Class 11 Chemistry Chapter 9 - Hydrocarbons - 2025-26

CBSE Notes Class 11 Chemistry Chapter 5 - Thermodynamics - 2025-26

CBSE Notes Class 11 Chemistry Chapter 8 - Organic Chemistry Some Basic Principles And Techniques - 2025-26

JEE Advanced Weightage 2025 Chapter-Wise for Physics, Maths and Chemistry




