The value for crystal field stabilisation energy is zero for:
(A) ${ K }_{ 2 }\left[ Mn{ F }_{ 6 } \right]$
(B) ${ K }_{ 3 }\left[ Fe(CN)_{ 6 } \right]$
(C) ${ K }_{ 3 }\left[ Fe{ F }_{ 6 } \right]$
(D) ${ K }_{ 4 }\left[ Fe(CN)_{ 6 } \right]$
Answer
266.7k+ views
Hint: Crystal field stabilization energy: Crystal field stabilization energy is the energy of electronic configuration in the ligand field - Electronic configuration in the isotropic field.
Complete answer step-by-step:
> The splitting pattern in each complex compound is octahedral. If a strong field ligand is present, then back pairing will take place as in the case of B and D, and splitting energy can never be zero, since no electron can go into ${ e }_{ g }$ orbitals.
But if a strong field is not present, then back pairing will not take place as in the case of A and C.
> The splitting pattern in ${ K }_{ 3 }\left[ Fe{ F }_{ 6 } \right]$ is;
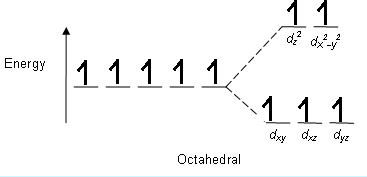
Atomic number of Fe = ${ 26 }$
Electronic configuration = $\left[ Ar \right] { 3d }^{ 6 }{ 4s }^{ 2 }$
For, ${ Fe }^{ +3 } = \left[ Ar \right] { 3d }^{ 5 } or { t }_{ 2g }^{ 3 }{ e }_{ g }^{ 2 }$
The crystal field splitting energy for octahedral complexes are given by;
CFSE = ${ -0.4\times }{ t }_{ 2g }{ +0.6\times }{ e }_{ g }$
By putting the values, we get
CFSE = ${ -0.4\times 3 }{ +0.6\times 2 }$
CFSE = 0
Now, for ${ K }_{ 2 }\left[ Mn{ F }_{ 6 } \right]$
Atomic number of Mn = ${ 25 }$
Electronic configuration = $\left[ Ar \right] { 3d }^{ 5 }{ 4s }^{ 2 }$
For, ${ Mn }^{ +3 } = \left[ Ar \right] { 3d }^{ 4 }or { t }_{ 2g }^{ 3 }{ e }_{ g }^{ 1 }$
The crystal field splitting energy for octahedral complexes are given by;
CFSE = ${ -0.4\times }{ t }_{ 2g }{ +0.6\times }{ e }_{ g }$
By putting the values, we get
CFSE = ${ -0.4\times 3 }{ +0.6\times 1 }$
CFSE = ${ -0.6 } \Delta _{ 0 }$
Hence, we can say that the value for crystal field stabilization energy is zero for ${ K }_{ 3 }\left[ Fe{ F }_{ 6 } \right]$.
The correct option is C.
Note: The possibility to make a mistake is that you may use the crystal field splitting energy formula for tetrahedral, not octahedral. But all the compounds are octahedral so do not confuse between them.
Complete answer step-by-step:
> The splitting pattern in each complex compound is octahedral. If a strong field ligand is present, then back pairing will take place as in the case of B and D, and splitting energy can never be zero, since no electron can go into ${ e }_{ g }$ orbitals.
But if a strong field is not present, then back pairing will not take place as in the case of A and C.
> The splitting pattern in ${ K }_{ 3 }\left[ Fe{ F }_{ 6 } \right]$ is;

Atomic number of Fe = ${ 26 }$
Electronic configuration = $\left[ Ar \right] { 3d }^{ 6 }{ 4s }^{ 2 }$
For, ${ Fe }^{ +3 } = \left[ Ar \right] { 3d }^{ 5 } or { t }_{ 2g }^{ 3 }{ e }_{ g }^{ 2 }$
The crystal field splitting energy for octahedral complexes are given by;
CFSE = ${ -0.4\times }{ t }_{ 2g }{ +0.6\times }{ e }_{ g }$
By putting the values, we get
CFSE = ${ -0.4\times 3 }{ +0.6\times 2 }$
CFSE = 0
Now, for ${ K }_{ 2 }\left[ Mn{ F }_{ 6 } \right]$
Atomic number of Mn = ${ 25 }$
Electronic configuration = $\left[ Ar \right] { 3d }^{ 5 }{ 4s }^{ 2 }$
For, ${ Mn }^{ +3 } = \left[ Ar \right] { 3d }^{ 4 }or { t }_{ 2g }^{ 3 }{ e }_{ g }^{ 1 }$
The crystal field splitting energy for octahedral complexes are given by;
CFSE = ${ -0.4\times }{ t }_{ 2g }{ +0.6\times }{ e }_{ g }$
By putting the values, we get
CFSE = ${ -0.4\times 3 }{ +0.6\times 1 }$
CFSE = ${ -0.6 } \Delta _{ 0 }$
Hence, we can say that the value for crystal field stabilization energy is zero for ${ K }_{ 3 }\left[ Fe{ F }_{ 6 } \right]$.
The correct option is C.
Note: The possibility to make a mistake is that you may use the crystal field splitting energy formula for tetrahedral, not octahedral. But all the compounds are octahedral so do not confuse between them.
Recently Updated Pages
States of Matter Chapter For JEE Main Chemistry

Classification of Drugs in Chemistry: Types, Examples & Exam Guide

Types of Solutions in Chemistry: Explained Simply

Difference Between Alcohol and Phenol: Structure, Tests & Uses

[Awaiting the three content sources: Ask AI Response, Competitor 1 Content, and Competitor 2 Content. Please provide those to continue with the analysis and optimization.]

Sign up for JEE Main 2026 Live Classes - Vedantu

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




