The shape of $PC{{l}_{3}}$ molecule is:
A. trigonal bipyramid
B. tetrahedral
C. pyramidal
D. square planar
Answer
263.7k+ views
Hint: $PC{{l}_{3}}$ is called phosphorus trichloride, which is formed from phosphorus and chlorine. It is actually a very toxic liquid which when reacted with water releases HCl gas. The hybridisation in $PC{{l}_{3}}$is $s{{p}^{3}}$.
Step by step solution:
- Now let’s write the electronic configuration of phosphorus,
The atomic number of phosphorus is 15, so we can write electronic configuration as –
\[1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}3{{s}^{2}}3{{p}^{3}}\]
- Here, if we have to find the hybridisation, then it is equal to the sum of the number of sigma bonds and the number of lone pairs.
- Here, we can see that all bonds are of (P-Cl) type, we can see from the diagram drawn below that there are three P-Cl single bonds present, so the number of sigma bonds will be 3.
- And we can see here that there are 5 electrons present in valence shell that is $3{{s}^{2}}3{{p}^{3}}$, so ; two electrons are not participating in pairing , therefore the number of lone pairs will be equal to 1.
- So, we can say that hybridisation in $PC{{l}_{3}}$is $s{{p}^{3}}$.
- Structure of $PC{{l}_{3}}$ is:
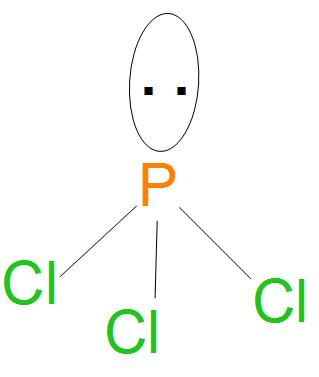
- Hence, from the structure and hybridisation of $PC{{l}_{3}}$ ,we can say that the geometry is pyramidal.
Additional information:
- $PC{{l}_{3}}$ is having a bond angle of approximately .
- $PC{{l}_{3}}$ is found to produce , which are used in many applications, including herbicides, insecticides etc.
- It is also found to be used directly as a reagent in organic synthesis, which is also used to convert many primary as well as secondary alcohols into alkyl chlorides.
Note:
We should not get confused in between $PC{{l}_{3}}$ and $PC{{l}_{5}}$. $PC{{l}_{3}}$ is phosphorus trichloride and is having pyramidal geometry, whereas $PC{{l}_{5}}$ is phosphorus pentachloride that has trigonal bipyramidal geometry.
Step by step solution:
- Now let’s write the electronic configuration of phosphorus,
The atomic number of phosphorus is 15, so we can write electronic configuration as –
\[1{{s}^{2}}2{{s}^{2}}2{{p}^{6}}3{{s}^{2}}3{{p}^{3}}\]
- Here, if we have to find the hybridisation, then it is equal to the sum of the number of sigma bonds and the number of lone pairs.
- Here, we can see that all bonds are of (P-Cl) type, we can see from the diagram drawn below that there are three P-Cl single bonds present, so the number of sigma bonds will be 3.
- And we can see here that there are 5 electrons present in valence shell that is $3{{s}^{2}}3{{p}^{3}}$, so ; two electrons are not participating in pairing , therefore the number of lone pairs will be equal to 1.
- So, we can say that hybridisation in $PC{{l}_{3}}$is $s{{p}^{3}}$.
- Structure of $PC{{l}_{3}}$ is:

- Hence, from the structure and hybridisation of $PC{{l}_{3}}$ ,we can say that the geometry is pyramidal.
Additional information:
- $PC{{l}_{3}}$ is having a bond angle of approximately .
- $PC{{l}_{3}}$ is found to produce , which are used in many applications, including herbicides, insecticides etc.
- It is also found to be used directly as a reagent in organic synthesis, which is also used to convert many primary as well as secondary alcohols into alkyl chlorides.
Note:
We should not get confused in between $PC{{l}_{3}}$ and $PC{{l}_{5}}$. $PC{{l}_{3}}$ is phosphorus trichloride and is having pyramidal geometry, whereas $PC{{l}_{5}}$ is phosphorus pentachloride that has trigonal bipyramidal geometry.
Recently Updated Pages
JEE Main Mock Test 2025-26: Principles Related To Practical

JEE Main 2025-26 Experimental Skills Mock Test – Free Practice

JEE Main 2025-26 Electronic Devices Mock Test: Free Practice Online

JEE Main 2025-26 Mock Tests: Free Practice Papers & Solutions

JEE Main 2025-26: Magnetic Effects of Current & Magnetism Mock Test

JEE Main Statistics and Probability Mock Test 2025-26

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

NCERT Solutions For Class 11 Chemistry In Hindi Chapter 1 Some Basic Concepts Of Chemistry - 2025-26

How to Convert a Galvanometer into an Ammeter or Voltmeter

Understanding Electromagnetic Waves and Their Importance




