The propene reacts with \[HBr\] to form
(a) Ethane
(b) Hexane
(c) 1-bromo-propane
(d) 2-bromo propane
Answer
267.6k+ views
Hint: Propene is a member of alkene compound. It is very reactive due to the presence of an unsymmetrical double bond. Propene readily reacts with \[HBr\] and forms bromide compounds. The reaction of \[HBr\] with propene is an example of a nucleophilic addition reaction.
Complete Step by Step Answer:
Propene is an example of an unsymmetrical alkene in which three carbon atoms are present.
Propene readily reacts with \[HBr\]and forms 2-bromopropane (major product) and 1-bromopropane (minor product).
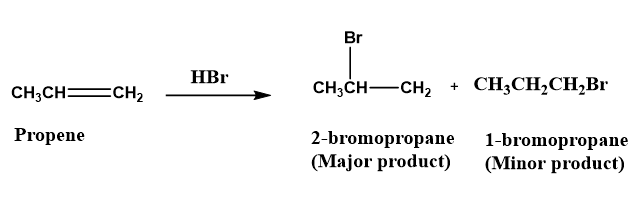
Image: Synthesis of 2-bromopropane
For the addition of \[HBr\]on an unsymmetrical alkene Marconikov rule is used to find a location at which the carbon atom of double bond the hydrogen and bromine atoms undergoes the addition.
According to the Marconikov rule the hydrogen atom of \[HBr\] prefers to bind the end of the carbon-carbon double bond which possesses the highest number of the hydrogen atom. Whereas the bromine atom prefers to bind the end of the carbon-carbon double bond which possesses the lowest number of hydrogen atoms.
Mechanism:
\[HBr\]is a polar molecule due to the electronegativity difference between hydrogen and bromine atoms.
Due to its polar nature, the \[HBr\]molecule can dissociate into \[{H^ + }\] (electrophile) and \[B{r^ - }\](nucleophile) when it approaches the alkene.
The electrophile (\[{H^ + }\]) can attack the double bond of propene and it can form a primary and secondary carbocation. Because secondary carbocation is more stable than primary carbocation. Therefore, formation of secondary carbocation is observed.
After the addition of electrophile (\[{H^ + }\]) the bromide ion or nucleophile (\[B{r^ - }\]) react with formed carbocation and forms the final product i.e., 2-bromopropane (as a major product).

Image: Mechanism of the addition of hydrobromic acid over an unsymmetrical alkene.
Therefore, from the above discussion, it is quite clear that option (d) will be the correct answer.
Note: When the addition of \[HBr\]to the unsymmetrical alkene takes place in the presence of peroxide then this reaction is called as anti-Markovnikov rule or peroxide effect or sometimes it is also known as the Kharasch effect. In such cases, the major product is derived from less stable carbocation.
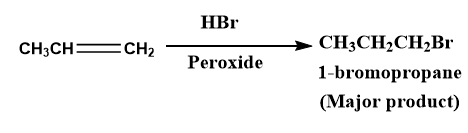
Image: addition of hydrobromic acid on alkene in the presence of peroxide.
Complete Step by Step Answer:
Propene is an example of an unsymmetrical alkene in which three carbon atoms are present.
Propene readily reacts with \[HBr\]and forms 2-bromopropane (major product) and 1-bromopropane (minor product).

Image: Synthesis of 2-bromopropane
For the addition of \[HBr\]on an unsymmetrical alkene Marconikov rule is used to find a location at which the carbon atom of double bond the hydrogen and bromine atoms undergoes the addition.
According to the Marconikov rule the hydrogen atom of \[HBr\] prefers to bind the end of the carbon-carbon double bond which possesses the highest number of the hydrogen atom. Whereas the bromine atom prefers to bind the end of the carbon-carbon double bond which possesses the lowest number of hydrogen atoms.
Mechanism:
\[HBr\]is a polar molecule due to the electronegativity difference between hydrogen and bromine atoms.
Due to its polar nature, the \[HBr\]molecule can dissociate into \[{H^ + }\] (electrophile) and \[B{r^ - }\](nucleophile) when it approaches the alkene.
The electrophile (\[{H^ + }\]) can attack the double bond of propene and it can form a primary and secondary carbocation. Because secondary carbocation is more stable than primary carbocation. Therefore, formation of secondary carbocation is observed.
After the addition of electrophile (\[{H^ + }\]) the bromide ion or nucleophile (\[B{r^ - }\]) react with formed carbocation and forms the final product i.e., 2-bromopropane (as a major product).

Image: Mechanism of the addition of hydrobromic acid over an unsymmetrical alkene.
Therefore, from the above discussion, it is quite clear that option (d) will be the correct answer.
Note: When the addition of \[HBr\]to the unsymmetrical alkene takes place in the presence of peroxide then this reaction is called as anti-Markovnikov rule or peroxide effect or sometimes it is also known as the Kharasch effect. In such cases, the major product is derived from less stable carbocation.

Image: addition of hydrobromic acid on alkene in the presence of peroxide.
Recently Updated Pages
Algebra Made Easy: Step-by-Step Guide for Students

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

JEE Energetics Important Concepts and Tips for Exam Preparation

Chemical Properties of Hydrogen - Important Concepts for JEE Exam Preparation

JEE General Topics in Chemistry Important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

How to Convert a Galvanometer into an Ammeter or Voltmeter

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 3 Chemical Kinetics - 2025-26




