The monobasic acid among the following is-
[A] ${{H}_{3}}P{{O}_{3}}$
[B] ${{H}_{2}}{{S}_{2}}{{O}_{7}}$
[C] ${{H}_{3}}P{{O}_{2}}$
[D] ${{H}_{2}}{{P}_{2}}{{O}_{7}}$
Answer
267.9k+ views
HINT: On ionisation with water the acids which produce a single hydronium ion i.e. readily ionises only one proton, are known as monobasic acids. The basicity/acidity of oxo-acids is defined by the number of –OH bonds present in the acid.
Complete step by step solution:
> We know acids produce hydronium ions (${{H}_{3}}{{O}^{+}}$) on ionisation. The number of hydronium ions produced makes it mono, di or tri-basic.
To find out if the given acid is monobasic tribasic or dibasic, we can also look at the number of –OH bonds in the given acid. If the acid contains one, two or three –OH bonds, they are monobasic, dibasic and tribasic respectively. To find out the number of hydroxyl groups present, we need to draw the structures of each of the given compound-
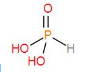
${{H}_{3}}P{{O}_{3}}$, phosphorous acid has two P-OH bonds and one P-O bond.
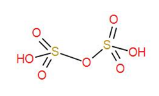
${{H}_{2}}{{S}_{2}}{{O}_{7}}$, disulphuric acid, also known as oleum has one S-O-S bond and four S-O bonds and two S-OH bonds.
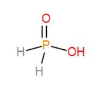
${{H}_{3}}P{{O}_{2}}$, hypo phosphorous acid has one P-O bond and one P-OH bond.
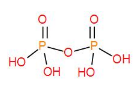
Lastly, ${{H}_{2}}{{P}_{2}}{{O}_{7}}$have one P-O-P bond and two P-O bonds.
> Here, ${{H}_{3}}P{{O}_{3}}$ is not tribasic as the –H atom present is the acidic hydrogen, it can readily ionize 2 protons (two –OH bonds present here) therefore it is a dibasic acid. Similarly, ${{H}_{2}}{{S}_{2}}{{O}_{7}}$ and ${{H}_{2}}{{P}_{2}}{{O}_{7}}$are dibasic and tribasic respectively.
Since ${{H}_{3}}P{{O}_{2}}$has only one proton available to ionize i.e. only one ${{H}^{+}}$can be donated, therefore it is monobasic.
Therefore, option [C] is the correct answer.
Additional information: According to the Bronsted-Lowrey concept, an acid is a substance that can release a proton and a base is a substance that can accept protons.
Note: It is important to remember here that the presence of –H atoms does not define the acidity of oxo-acids. We get the measure of acidity/basicity by the –OH bonds. It is also important to remember the structures of these common compounds as we often miss out the double bonded O atoms and write them as –OH instead.
Complete step by step solution:
> We know acids produce hydronium ions (${{H}_{3}}{{O}^{+}}$) on ionisation. The number of hydronium ions produced makes it mono, di or tri-basic.
To find out if the given acid is monobasic tribasic or dibasic, we can also look at the number of –OH bonds in the given acid. If the acid contains one, two or three –OH bonds, they are monobasic, dibasic and tribasic respectively. To find out the number of hydroxyl groups present, we need to draw the structures of each of the given compound-

${{H}_{3}}P{{O}_{3}}$, phosphorous acid has two P-OH bonds and one P-O bond.
${{H}_{2}}{{S}_{2}}{{O}_{7}}$, disulphuric acid, also known as oleum has one S-O-S bond and four S-O bonds and two S-OH bonds.

${{H}_{3}}P{{O}_{2}}$, hypo phosphorous acid has one P-O bond and one P-OH bond.

Lastly, ${{H}_{2}}{{P}_{2}}{{O}_{7}}$have one P-O-P bond and two P-O bonds.

> Here, ${{H}_{3}}P{{O}_{3}}$ is not tribasic as the –H atom present is the acidic hydrogen, it can readily ionize 2 protons (two –OH bonds present here) therefore it is a dibasic acid. Similarly, ${{H}_{2}}{{S}_{2}}{{O}_{7}}$ and ${{H}_{2}}{{P}_{2}}{{O}_{7}}$are dibasic and tribasic respectively.
Since ${{H}_{3}}P{{O}_{2}}$has only one proton available to ionize i.e. only one ${{H}^{+}}$can be donated, therefore it is monobasic.
Therefore, option [C] is the correct answer.
Additional information: According to the Bronsted-Lowrey concept, an acid is a substance that can release a proton and a base is a substance that can accept protons.
Note: It is important to remember here that the presence of –H atoms does not define the acidity of oxo-acids. We get the measure of acidity/basicity by the –OH bonds. It is also important to remember the structures of these common compounds as we often miss out the double bonded O atoms and write them as –OH instead.
Recently Updated Pages
Algebra Made Easy: Step-by-Step Guide for Students

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

JEE Energetics Important Concepts and Tips for Exam Preparation

Chemical Properties of Hydrogen - Important Concepts for JEE Exam Preparation

JEE General Topics in Chemistry Important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

How to Convert a Galvanometer into an Ammeter or Voltmeter

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Percentile vs Marks 2026: JEE Main Cutoff, AIR & IIT Admission Guide

JEE Advanced 2026 Marks vs Rank: Estimate IIT Rank from Your Score

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics

NCERT Solutions For Class 11 Chemistry In Hindi Chapter 1 Some Basic Concepts Of Chemistry - 2025-26

Understanding Electromagnetic Waves and Their Importance




