The final product C, obtained in the reaction, would be:
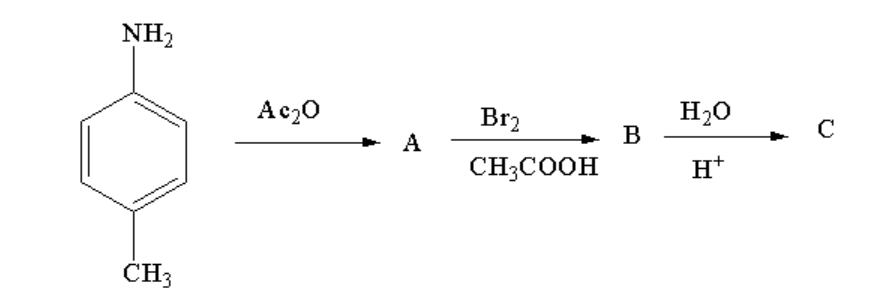
A. 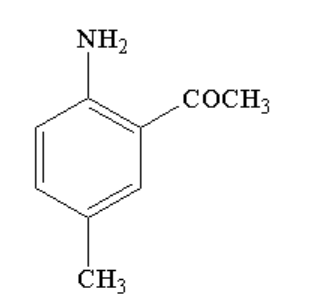
B. 
C. 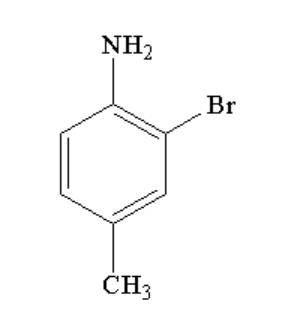
D. 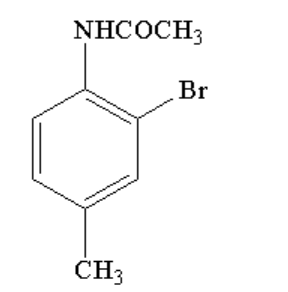
Answer
256.8k+ views
Hint: \[A{c_2}O\] stands for acetic anhydride which is used as a acetylating reagent in the first step. In the second step bromination takes place with bromine in acetic acid and in the third step hydrolysis takes place.
Complete step by step solution:By looking in the reaction sequence, it can be seen that the reaction is taking place in three steps.
In the first step p-toluidine reacts with acetic anhydride to form an acetylated compound known as N-p-tolylacetamide. Acetylation is a reaction where an acetyl group is introduced to the reactant. In the first step, amine is getting converted to amide.
In the second step, p-tolyl acetamide reacts with bromine in presence of acetic acid to form N-(2-bromo-4-methylphenyl)acetamide. The reaction is known as bromination reaction where bromine is added to the ortho position of the reactant.
In the third step, N-(2-bromo-4-methylphenyl)acetamide undergoes hydrolysis to form 2-bromo-4-methylbenzenamine. Hydrolysis is a reaction where one of the reactants is water or in other way a water molecule is added to the reactant. Here, the amide group is again converted to amine group.
The overall reaction for the conversion is shown below.
Image: Reaction sequence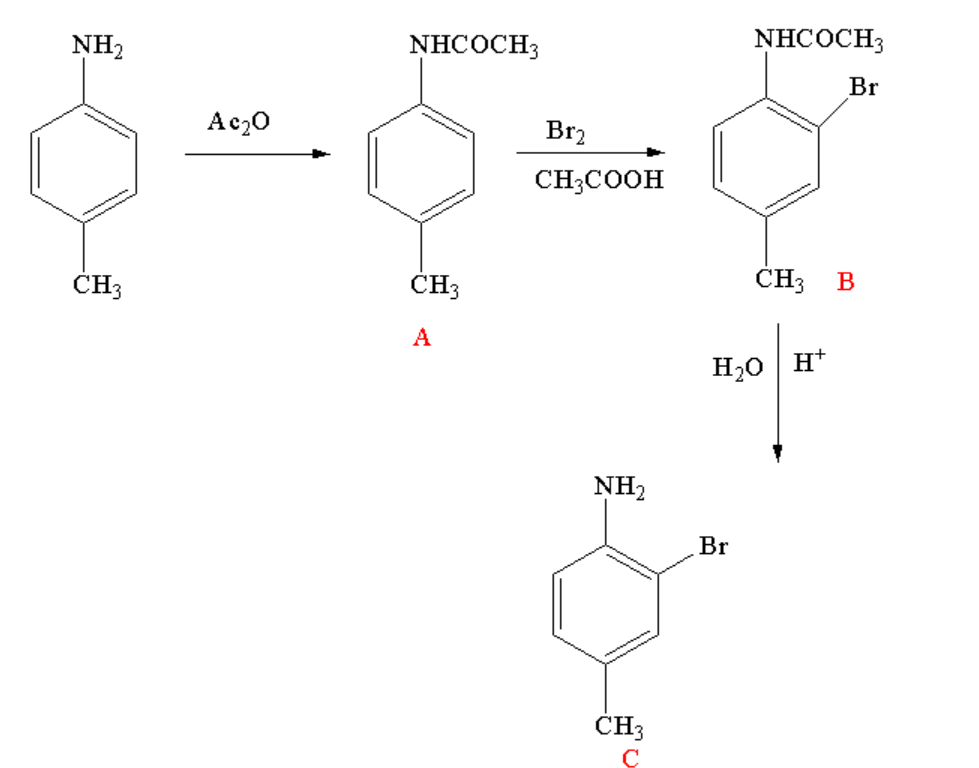
Image: Reaction sequence
Therefore, option C is correct.
Note: The common name of N-(2-bromo-4-methylphenyl)acetamide is bromoacetanilide. The final compound formed is a monobromo compound as only one bromine atom is attached. It should be noted that acetic acid in bromination is used as a solvent.
Complete step by step solution:By looking in the reaction sequence, it can be seen that the reaction is taking place in three steps.
In the first step p-toluidine reacts with acetic anhydride to form an acetylated compound known as N-p-tolylacetamide. Acetylation is a reaction where an acetyl group is introduced to the reactant. In the first step, amine is getting converted to amide.
In the second step, p-tolyl acetamide reacts with bromine in presence of acetic acid to form N-(2-bromo-4-methylphenyl)acetamide. The reaction is known as bromination reaction where bromine is added to the ortho position of the reactant.
In the third step, N-(2-bromo-4-methylphenyl)acetamide undergoes hydrolysis to form 2-bromo-4-methylbenzenamine. Hydrolysis is a reaction where one of the reactants is water or in other way a water molecule is added to the reactant. Here, the amide group is again converted to amine group.
The overall reaction for the conversion is shown below.
Image: Reaction sequence

Image: Reaction sequence
Therefore, option C is correct.
Note: The common name of N-(2-bromo-4-methylphenyl)acetamide is bromoacetanilide. The final compound formed is a monobromo compound as only one bromine atom is attached. It should be noted that acetic acid in bromination is used as a solvent.
Recently Updated Pages
Electricity and Magnetism Explained: Key Concepts & Applications

JEE Energetics Important Concepts and Tips for Exam Preparation

JEE Isolation, Preparation and Properties of Non-metals Important Concepts and Tips for Exam Preparation

JEE Main 2023 (February 1st Shift 1) Maths Question Paper with Answer Key

JEE Main 2023 (February 1st Shift 2) Maths Question Paper with Answer Key

JEE Main 2023 (February 1st Shift 2) Chemistry Question Paper with Answer Key

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

JEE Main 2026 Application Login: Direct Link, Registration, Form Fill, and Steps

JEE Main Colleges 2026: Complete List of Participating Institutes

JEE Main Marking Scheme 2026- Paper-Wise Marks Distribution and Negative Marking Details

Hybridisation in Chemistry – Concept, Types & Applications

Other Pages
JEE Advanced 2026 - Exam Date (Released), Syllabus, Registration, Eligibility, Preparation, and More

CBSE Class 12 Chemistry Question Paper 2026 PDF Download (All Sets) with Answer Key

NCERT Solutions For Class 12 Chemistry Chapter 10 Biomolecules - 2025-26

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

NCERT Solutions For Class 12 Chemistry Chapter 2 Electrochemistry - 2025-26

NCERT Solutions For Class 12 Chemistry Chapter 1 Solutions - 2025-26




