The degrees of freedom of a diatomic gas at normal temperature is:
A) $3$
B) $4$
C) $5$
D) $6$
Answer
262.8k+ views
Hint: The degrees of freedom is the change in the state of the physical system or of the motion exhibited by the particular molecule which is located in a particular space. It varies for the monatomic, diatomic and the triatomic molecules.
Complete step by step solution:
The movement of the particle in the three dimensional space constitutes the three degrees of freedom. In the diatomic molecule, the whole molecule exhibits three degrees of freedom due to its centre of mass and also has another three degrees of freedom. These diatomic sets have been decomposed mainly in terms of the translation, vibration and the rotation of the molecule.
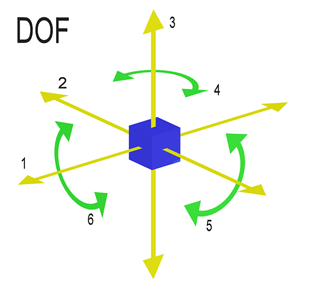
A single diatomic molecule has two rotational types of the motion and one vibrational type of the motion. In the diatomic molecule, the rotation takes place perpendicular to that of the axis joining the two atoms but it is not a physical degree of the rotation. Hence the degrees of freedom of the diatomic molecule is $6$ , but the vibration does not take place at the normal temperature. Hence the total number of the degree of freedom is calculated as follows.
$f = 3 + 2$
By adding the above degrees of freedom,
$f = 5$
Hence the degrees of freedom obtained for a diatomic gas molecule at a normal temperature is $5$ .
Thus the option (C) is correct.
Note: If the $N$ is the number of gas molecules in the container, hence the degrees of freedom is $f = 3N$ . If there occur any restrictions of the motion of the gas molecules and it is considered as $q$, then the degrees of freedom becomes $f = 3N - q$.
Complete step by step solution:
The movement of the particle in the three dimensional space constitutes the three degrees of freedom. In the diatomic molecule, the whole molecule exhibits three degrees of freedom due to its centre of mass and also has another three degrees of freedom. These diatomic sets have been decomposed mainly in terms of the translation, vibration and the rotation of the molecule.

A single diatomic molecule has two rotational types of the motion and one vibrational type of the motion. In the diatomic molecule, the rotation takes place perpendicular to that of the axis joining the two atoms but it is not a physical degree of the rotation. Hence the degrees of freedom of the diatomic molecule is $6$ , but the vibration does not take place at the normal temperature. Hence the total number of the degree of freedom is calculated as follows.
$f = 3 + 2$
By adding the above degrees of freedom,
$f = 5$
Hence the degrees of freedom obtained for a diatomic gas molecule at a normal temperature is $5$ .
Thus the option (C) is correct.
Note: If the $N$ is the number of gas molecules in the container, hence the degrees of freedom is $f = 3N$ . If there occur any restrictions of the motion of the gas molecules and it is considered as $q$, then the degrees of freedom becomes $f = 3N - q$.
Recently Updated Pages
Circuit Switching vs Packet Switching: Key Differences Explained

Dimensions of Pressure in Physics: Formula, Derivation & SI Unit

JEE Extractive Metallurgy Important Concepts and Tips for Exam Preparation

JEE Atomic Structure and Chemical Bonding important Concepts and Tips

JEE Amino Acids and Peptides Important Concepts and Tips for Exam Preparation

Electricity and Magnetism Explained: Key Concepts & Applications

Trending doubts
JEE Main 2026: Exam Dates, Session 2 Updates, City Slip, Admit Card & Latest News

JEE Main Participating Colleges 2026 - A Complete List of Top Colleges

Hybridisation in Chemistry – Concept, Types & Applications

Understanding the Electric Field of a Uniformly Charged Ring

Derivation of Equation of Trajectory Explained for Students

Understanding Atomic Structure for Beginners

Other Pages
JEE Advanced 2026 Notification Out with Exam Date, Registration (Extended), Syllabus and More

JEE Advanced Marks vs Ranks 2025: Understanding Category-wise Qualifying Marks and Previous Year Cut-offs

CBSE Notes Class 11 Physics Chapter 1 - Units And Measurements - 2025-26

NCERT Solutions For Class 11 Physics Chapter 1 Units And Measurements - 2025-26

Important Questions For Class 11 Physics Chapter 1 Units and Measurement - 2025-26

JEE Advanced Weightage Chapter Wise 2026 for Physics, Chemistry, and Mathematics




